Identification of targetable epitope surfaces from the high resolution structure of the superantigen Staphylococcal Enterotoxin L
Identification of targetable epitope surfaces from the high resolution structure of the superantigen Staphylococcal Enterotoxin L
Marino, S. F.; Siewert, K.; Curato, C.; Klamm, E.
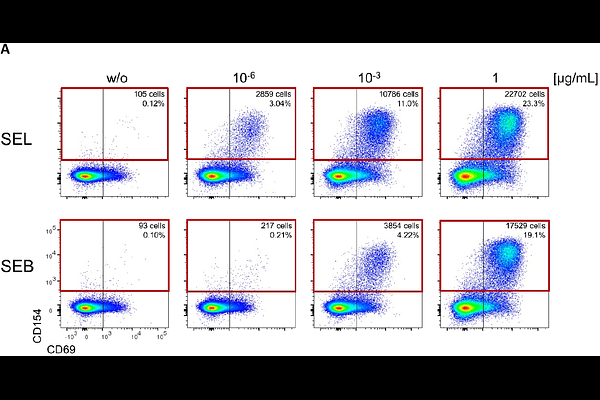
AbstractEmetic exotoxins secreted by Staphylococcus species (Staphylococcal enterotoxins, SEs) are a major cause of food poisoning cases worldwide and many are additionally classified as superantigens - able to potently activate T cells in an antigen independent manner. Fewer than half of the gene products of the known SE genes have been extensively characterized. The gene for Staphylococcal enterotoxin L (SEL) occurs in both foodborne and clinical isolates but no detailed structural characterization has yet been available. We report here the crystal structure of SEL and confirm its function as a superantigen via direct T cell activation assays. By comparison of the SEL sequence with that of its four closest homologues (SEI, SEK, SEM and SEQ), we have identified binding epitopes unique for SEL and mapped these regions onto the structure. These data provide the first high resolution view of SEL and the basis for the development of diagnostic procedures for its specific detection.