H3 dopaminylation and CaMKII modulate diffuse midline glioma response to CDK9 inhibition
H3 dopaminylation and CaMKII modulate diffuse midline glioma response to CDK9 inhibition
Murdaugh, R. L.; Eberl, B. R.; Richard, R. U.; Campos-Hensley, E. I.; Nnyagu, A. N.; Elam, W. A.; Tsao, A.-N. N.; Tremblay, J. R.; Ma, R.; Dei-Ampeh, A. K.; Pham, K.; Kraushaar, D. C.; Yu, K.; Olson, C. M.; Serin Harmanci, A.; Deneen, B.; Anastas, J. N.
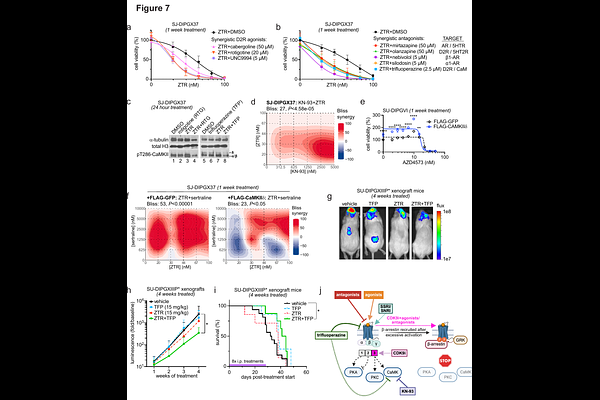
AbstractAberrant neurotransmitter signaling and transcriptional dysregulation are hallmarks of gliomagenesis and represent potential therapeutic targets. Monoamine neurotransmitters such as dopamine and serotonin primarily activate GPCRs but can also function epigenetically as histone H3 covalent modifications. Here, we uncover mechanisms of crosstalk between monoamine neurotransmitter signaling, histone dopaminylation, and oncogenic RNA polymerase II (Pol2) transcription in diffuse midline glioma (DMG). We find that co-treatment with Pol2-targeting CDK9 inhibitors (CDK9i) and FDA-approved neuropsychiatric drugs, like selective serotonin reuptake inhibitors (SSRIs), synergistically reduces DMG growth. Mechanistically, CDK9i+SSRI treatment alters histone dopaminylation patterns and represses synaptic and neurodevelopmental gene transcription associated with CDK9i resistance. Further phospho-proteomic analyses show that CDK9i monotherapy activates pro-survival CaMKII signaling, which can be suppressed by co-treatment with neuromodulatory drugs. These studies establish roles for H3 dopaminylation and neurotransmitter signaling in DMG gene regulation and response to CDK9i, suggesting that monoamine neurotransmitter pathways may be exploited as a therapeutic strategy for DMG.