Faf2/Ubx2 potentiates p97/Cdc48 segregase activity via functional priming and reinforcement of the UT3 domain in Ufd1
Faf2/Ubx2 potentiates p97/Cdc48 segregase activity via functional priming and reinforcement of the UT3 domain in Ufd1
Ren, Y.; Zheng, Q.; Duan, Y.; Xu, Z.; Qu, Q.; Weng, Y.; Cui, S.; Yu, Y.; Pan, M.; Liu, L.
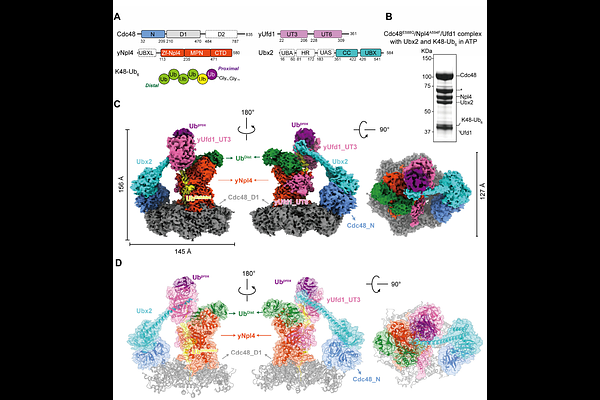
AbstractThe AAA+ segregase p97/Cdc48 extracts poly-ubiquitinated proteins from entrenched cellular environments to maintain proteostasis. However, its functional capacity is not uniform; extracting substrates from dense assemblies like stress granules or membrane necessitates a fully potentiated state. Here, using precisely synthesized ubiquitinated substrates, we show that the Faf2/Ubx2 cofactor hyperactivates p97/Cdc48, lowering its minimal ubiquitin chain requirement and broadening linkage specificity. Cryo-EM analysis of the activated complex demonstrates that Faf2/Ubx2 potentiates p97/Cdc48 segregase activity by structurally remodeling the substrate recognition module (K48-diUbProx) and priming its engagement with the AAA+ motor (p97/Cdc48-Npl4). These coordinated actions establish a 'pump-unit' architecture that drives efficient substrate processing. Our findings illuminate a conserved mechanism that unlocks the maximal capacity of p97/Cdc48 and provide a blueprint for therapeutically targeting its hyper-activated state in disease.