Exploring the mechanism of Panax Notoginseng in the treatment of skin wound based on network pharmacology and experimental verification
Exploring the mechanism of Panax Notoginseng in the treatment of skin wound based on network pharmacology and experimental verification
Li, Y.-b.; Li, Q.-l.; Liu, J.; Li, J.-c.; Geng, H.-m.; Li, G.-k.; Jin, C.; Luo, J.; Zhang, Z.
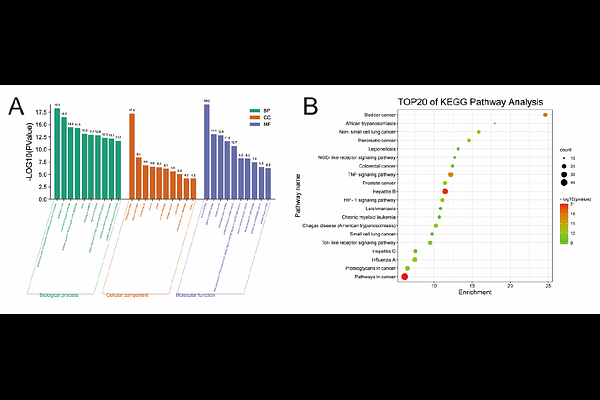
AbstractBackground How to shorten the healing cycle and reduce the incidence of infection is a difficult problem faced by clinicians. Panax notoginseng(PN), a traditional Chinese medicine, can promote the absorption of inflammatory exudates, granulation tissue formation and epidermal proliferation, effectively inhibit the inflammatory reaction of wounds and promote the healing of skin wounds, but its molecular mechanism has not been fully clarified so far. Based on network pharmacology and animal experiments, this study explored the target and molecular mechanism of PN in the treatment of skin wound. Methods Through network pharmacology, we screened the active components of PN and the common targets related to skin wounds, constructed a target protein-protein interaction (PPI) network, and performed GO and KEGG enrichment analysis. Using the MCODE and CytoHubba plugins, we explored core functional modules and key targets, ultimately constructing a visual network of PN components-targets-pathways. In the experimental section, Forty-eight male Sprague-Dawley (SD) rats were randomly divided into a control group and a PN group, with 24 rats in each group, and underwent full-thickness skin excision. Postoperatively, the PN group received intraperitoneal injections of drugs, while the control group received an equal amount of saline. Data were collected on postoperative days 1, 4, and 7, and hematoxylin and eosin (HE) staining, immunohistochemical staining, quantitative real-time polymerase chain reaction (qRT-PCR), and enzyme-linked immunosorbent assay (ELISA) were used to evaluate skin healing and detect changes in the expression of TNF-, IL-6, and IL-10 in the tissues. Results This study identified 8 major active components, 156 targets, and 115 signaling pathways involved in the treatment of skin wounds in rats using PN. The top 10 core target genes included TNF, IL-6, and IL-10, primarily enriched in signaling pathways such as NF-{kappa}B, MAPK, and JAK-STAT. Animal experiments revealed that at 4 and 7 days post-injury, the wound area in the PN group was significantly smaller than that in the control group (P<0.05). HE staining showed reduced infiltration of neutrophils and inflammatory cells in the injury area at 7 days in the PN group, accompanied by more pronounced fibroblast proliferation and collagen secretion. Molecular detection indicated that TNF-, IL-6, and IL-10 positive reactants were mainly distributed in the cytoplasm and matrix of epidermal cells, inflammatory cells, and fibroblasts in the skin. qRT-PCR and ELISA results showed that TNF- expression in the PN group was significantly lower than that in the control group at 4 and 7 days (P<0.01). IL-6 expression was lower than that in the control group at all time points, peaking at 4 days and then decreasing (P<0.01). IL-10 expression was significantly lower than that in the control group at 1 and 7 days (P<0.01). Conclusion PN treatment for skin wounds exhibits characteristics such as multi-component, multi-target, multi-pathway synergistic effects, and various regulatory pathways. It can reshape the dynamic balance of the cytokine network, optimize the temporal progression of "inflammation initiation - repair transition - tissue remodeling", and improve skin wound healing.