Deployment and transcriptional evaluation of nitisinone, an FDA-approved drug, to control bed bugs
Deployment and transcriptional evaluation of nitisinone, an FDA-approved drug, to control bed bugs
Sterkel, M.; Tompkin, J.; Schal, C.; Guera, L. M.; Pessoa, G. C. D.; Oliveira, P. L.; Benoit, J. B.
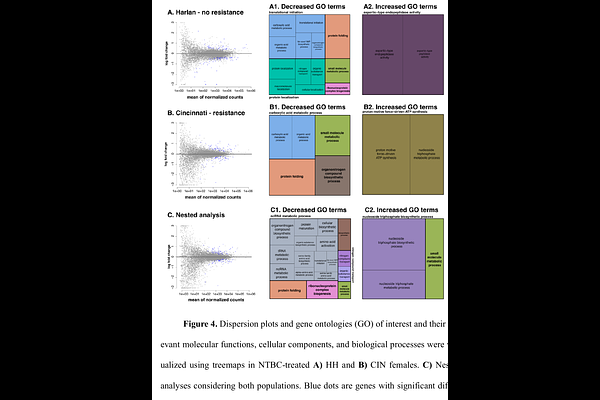
AbstractBed bugs are blood-feeders that rapidly proliferate into large indoor infesta-tions. Their bites can cause allergies, secondary infections and psychological stress, among other problems. Although several tactics for their management have been used, bed bugs continue to spread worldwide wherever humans reside. This is mainly due to human-mediated transport and their high resistance to several clas-ses of insecticides. New treatment options with novel modes of action are required for their control. In this study, we evaluated the use of nitisinone (NTBC), an FDA-approved drug, for bed bug control in an insecticide-susceptible (HH) and an insecticide-resistant (CIN) population. Although NTBC was lethal to both pop-ulations when administered orally or applied topically in very low doses, we ob-served a slight but significant resistance in the CIN population. Transcriptomic analysis in both populations indicated that NTBC treatment elicited a broad sup-pression of genes associated with RNA post-transcriptional modifications, transla-tion, endomembrane system, protein post-translational modifications and protein folding. The CIN population exhibited higher ATP production and xenobiotic de-toxification. Feeding studies on a mouse model highlight that NTBC could be used as a control method of bed bugs by host treatment. The results demonstrate that NTBC can be used as a new active ingredient for bed bug control by topical or oral treatment and shed light on the molecular mechanisms of suppressed tyro-sine metabolism following NTBC treatment.