A Unified Control of Cellular Differentiation: From Temporal Multistability to Spatial Pattern Formation in Gene Regulatory Networks
A Unified Control of Cellular Differentiation: From Temporal Multistability to Spatial Pattern Formation in Gene Regulatory Networks
Bansod, T.; Kaur, A.; Jolly, M. K.; Roy, U.
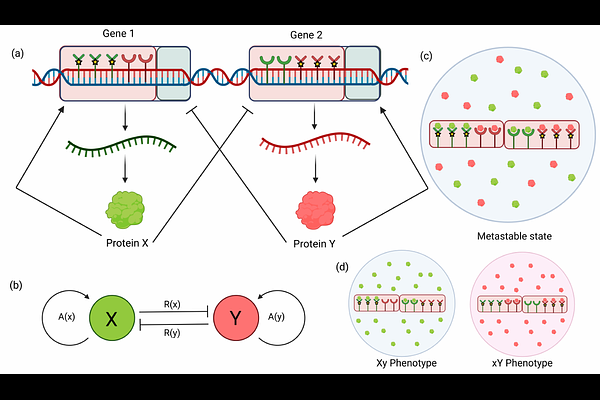
AbstractHow genetically identical cells spontaneously break symmetry to assume divergent fates is a fundamental problem in developmental biology. While modern genomics has mapped the vast molecular repertoire involved in gene regulation, understanding the mechanism of cell state transitions that drive differentiation remains a formidable challenge. To address this, we use a reaction-kinetic framework to analyze recurring motifs of two and three competing master regulators. While typically such circuits are studied numerically, we show that assuming symmetry in nodes and interactions provides exact analytical description of the bifurcations governing cell fate transitions. We find that the possible cell fates across all considered topologies are dictated by a single dimensionless quantity, {beta}-the ratio of protein degradation to production rates. In the binary Toggle Switch (TS), decreasing {beta} destabilizes the symmetric (stem cell) state, giving rise to two asymmetric (differentiated) fates via a supercritical pitchfork bifurcation. In the three-component Toggle Triad (TT), low values of {beta} yield three asymmetric fates through subcritical pitchfork bifurcation, creating an intermediate range of {beta} where both symmetric and asymmetric fates are simultaneously stable. For the Self-Activating Toggle Switch (SATS), we identify a new parameter for the self-activation threshold ({theta}) and show that decreasing {theta} progressively stabilizes the uncommitted state, leading to a regime of tristability. Building on these temporal bifurcations, we next address the feasibility of spatial structure formation: can these multistable fates stably coexist within a spatial domain? Through a minimal model of cell-cell communication via free diffusion, we extend these motifs into reaction-diffusion systems, which reveals a direct role of network topology on spatial organization. We prove that any heterogeneous pattern in two-node circuits is inherently transient and unstable. In contrast, the three-node repressive network supports the stable spatial coexistence of differentiated phenotypes through pure diffusion, a phenomenon we analyze by studying heteroclinic interface solutions as building blocks. By reducing complex regulatory dynamics to tractable models with physically meaningful parameters, we establish a minimal framework which relates topology to cell fate. Finally, the effects of temporal multistability on pattern formation provide an excellent studying ground for morphogenesis, synthetic biology, and the overarching problem of spatiotemporal self-organization.