The dynamics of introgression across an adaptive radiation: examining hybrid speciation and parallel adaptation in North American Vitis
The dynamics of introgression across an adaptive radiation: examining hybrid speciation and parallel adaptation in North American Vitis
Wang, T.; Fiscus, C.; Landis, J.; Cochetel, N.; Morales-Cruz, A.; Cantu, D.; Aguirre-Liguori, J. A.; Gaut, B. S.
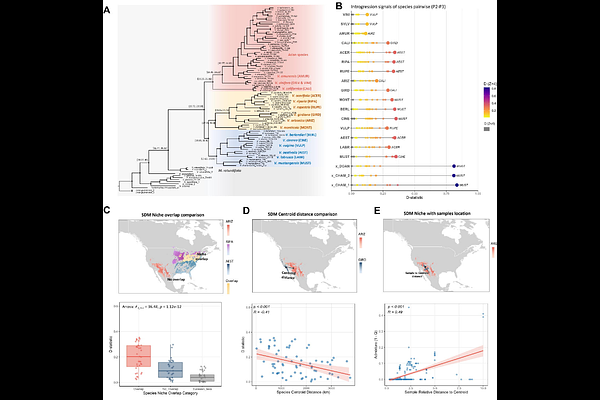
AbstractHybridization between species can contribute to parallel adaptive events and, in some cases, speciation. However, the prevalence of introgression is rarely measured across multiple species, meaning that it is rarely integrated with landscape-scale processes or connected to adaptive repeatability. Here, we analyzed whole-genome resequencing data from 639 accessions representing 48 Vitis species. Vitis is notable for the domesticated grapevine (V. vinifera), multiple economically important North American species, and as an example of a temperate adaptive radiation. Our dataset included population-level sampling for 19 species, from which we reconstructed individual- and species-level phylogenetic frameworks for the genus. The analyses uncovered widespread evidence of introgression, comprising ~14% of the average Vitis genome. Introgression was associated with geographic distribution between species, and highly admixed individuals were more frequently found near ecological niche margins. Genetic analyses further indicated that previously recognized hybrid taxa, V. x doaniana and V. x champinii, likely represent hybrid swarms rather than distinct species. We also assessed patterns of adaptive repeatability across eight species with denser population sampling. For six of the eight species, most of the detected sweeps overlapped with sweeps in other species; on average, more recently diverged species shared more overlapping sweeps. Parallel sweep events were driven mostly by introgression, although a substantial fraction (~40%) was attributed to selection on ancestral standing variation. By integrating population genomic data across many species, this study highlights the central role of shared genetic variation across an adaptive radiation.