Structural basis of the lobster carapace blue colour mediated by an HPR protein
Structural basis of the lobster carapace blue colour mediated by an HPR protein
Cedri, M. C.; Bansia, H.; Amici, A.; Ortore, M. G.; McCarthy, A.; Mueller-Dieckmann, C.; Raffaelli, N.; Durbeej, B.; Lingas, R.; Wang, T.; Des Georges, A.; Cianci, M.
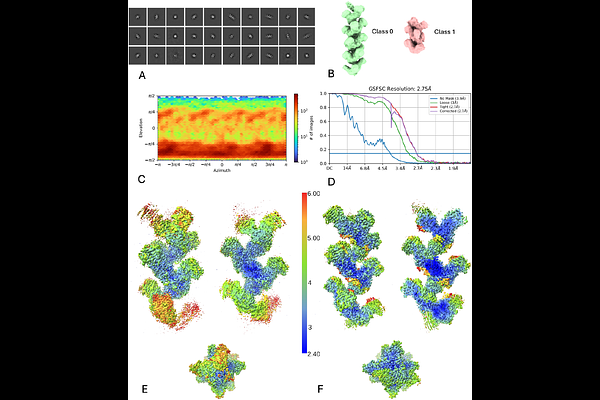
AbstractThe chemical basis underlying the striking blue hue of live H. americanus, known as American lobster, are studied in evolutionary biology and in polyene physical chemistry. Carapace colouration is generated by the antioxidant astaxanthin bound within the carotenoprotein crustacyanin complexes. Here, we present the ex vivo structure of the most abundant -crustacyanin and {beta}-crustacyanin forms, determined respectively by cryo-electron microscopy and X-ray crystallography to a resolution of 2.75 [A]. Our structural analysis reveals -crustacyanin as an elongated arrangement of {beta}-crustacyanin heterodimers tethered by an heptatricopeptide repeat (HPR) protein. In vitro complex formation between the {beta}-crustacyanin unit with a synthetic heptatricopeptide reproduces the observed blue colour of -crustacyanin, identifying the HPR protein, in concert with crustacyanins, as contributor in tuning carapace colour. Overall, these results explain how nature adjusts the colour across the entire visible spectrum by exploiting the bathochromic shift of astaxanthin from its unbound red form ({lambda}max = 472 nm) firstly to the {beta}-crustacyanin violet bound form ({lambda}max = 591 nm), and then to the -crustacyanin bound blue form ({lambda}max = 631 nm).