Engineered Allosteric RNA Editors Enable Compact, Stimulus-Responsive Post-Transcriptional Circuits
Engineered Allosteric RNA Editors Enable Compact, Stimulus-Responsive Post-Transcriptional Circuits
Marzilli, A. M.; Ngo, J. T.
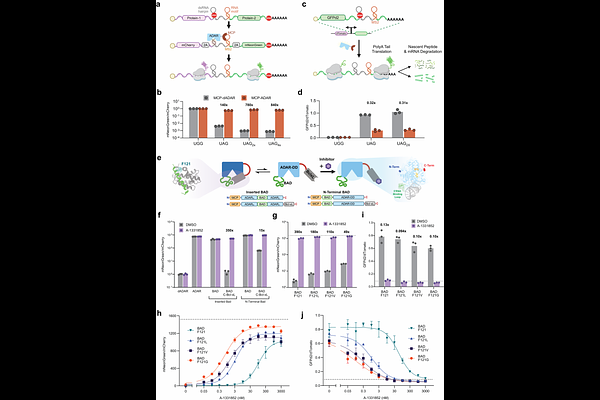
AbstractTranslational regulation offers a powerful biological control axis with the potential to enable programmable control over synthetic mRNAs. Here, we introduce inducible Deaminases Acting on RNA (iDARs): deaminase domains (DDs) with conditional RNA-editing activities. Using a domain-insertion strategy, we designed autoinhibited enzymes that can be converted into active RNA editors in response to triggers based on small molecules (chemiDARs), intracellular antigens (antiDARs), protease cleavage (lysiDAR), and optical excitation (optiDAR). Coupling these domains with novel stop codon containing RNA substrates enabled conditional protein translation or transcript degradation. Mutational tuning of inositol hexaphosphate (IP6)-binding pockets produced tightly regulated deaminases with minimal basal activity, facilitating dose-dependent readthrough translation in response to low-nanomolar drug concentration, with dynamic ranges exceeding 100-fold. By encoding iDARs alongside their substrates, we developed 'self-editing' polycistronic transcripts capable of directing translation of encoded proteins in a trigger-dependent manner following delivery to cells as in vitro transcribed mRNAs. Overall, iDARs provide a generalizable framework for generating controllable deaminases, enabling the design of post-transcriptional circuits that link biochemical sensing to readouts based on de novo translation or mRNA decay.