The HMD domain of the PAF complex primes Rad6-Bre1 E3 ligase complexes for H2B ubiquitination
The HMD domain of the PAF complex primes Rad6-Bre1 E3 ligase complexes for H2B ubiquitination
Tariq, A.; Ohsawa, S.; Zenezini Chiozzi, R.; Patsis, P.; Williams, C.; Stirpe, A.; Clarke, T. A.; Thalassinos, K.; Buehler, M.; Schalch, T.
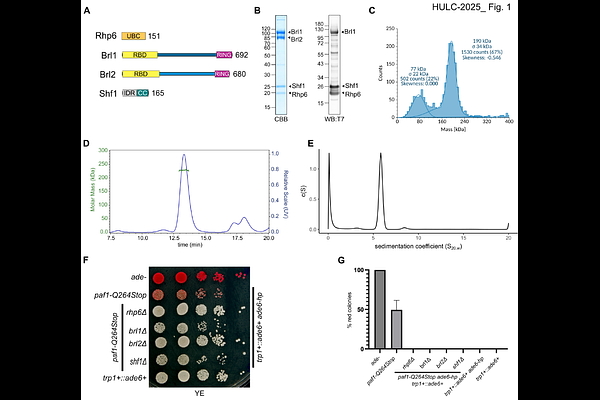
AbstractMono-ubiquitination of histone H2B (H2Bub) is deposited by Bre1-type E3 ubiquitin ligase complexes during transcription elongation and is critical for chromatin organization, DNA repair, and transcription regulation. In S. pombe, this activity is carried out by HULC, a complex of the Bre1 homologs Brl1 and Brl2 with Rhp6 and Shf1. While H2Bub deposition depends on recruitment of HULC to RNA Pol II by the Paf1 complex (PAF1C), the molecular basis for how PAF1C activates HULC has remained unclear. Using AlphaFold modelling, biochemical reconstitution, and functional assays, we define the architecture of HULC as a flexible 1:1:1:1 assembly in which Shf1 stabilizes an elongated coiled-coil hairpin that brings the RING and Rad6-binding domains into proximity. However, for full stimulation of HULC activity, we find that the HMD domain of the PAF1C subunit Prf1 stimulates ubiquitin transfer through a RING-binding region (RBR) that repositions the RING domains adjacent to Rhp6 in a catalytically competent configuration. These findings reveal an activation mechanism in which transcription-associated Prf1 primes HULC for ubiquitin transfer through the Prf1RBR, revealing a critical regulatory interface for H2B ubiquitination.