Bioimpedance-assisted characterization of cardiac electroporation and anisotropic homogenization by pulsed field ablation
Bioimpedance-assisted characterization of cardiac electroporation and anisotropic homogenization by pulsed field ablation
Jacobs, E. J.; Santos, P. P.; Parizi, S. S.; Dunham, S. N.; Davalos, R. V.
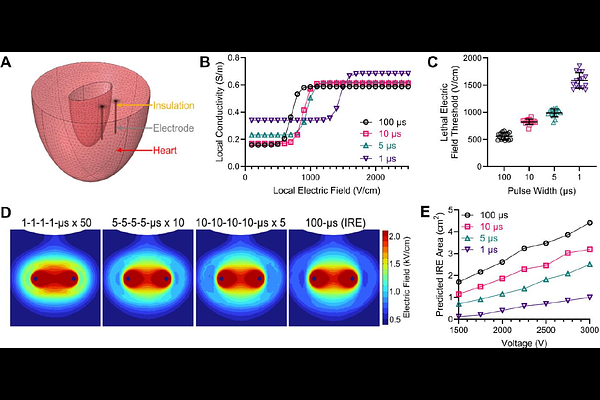
AbstractObjective: Pulsed field ablation (PFA) relies on irreversible electroporation to create nonthermal cardiac lesions, yet real-time indicators of electroporation progression and validated lethal electric field thresholds remain limited. This study aimed to develop a bioimpedance-based metric for real-time monitoring of cardiac electroporation, evaluate the impact of myocardial anisotropy under electroporation conditions, and derive waveform-specific lethal electric field thresholds. Introduction: Current PFA procedures lack direct intraoperative feedback on lesion formation, and uncertainty remains regarding the role of myocardial fiber orientation in shaping electric field distributions. Because electroporation dynamically alters tissue electrical properties, monitoring these changes during treatment may improve prediction of ablation outcomes. Methods: PFA was delivered to fresh ex vivo porcine ventricular tissue using clinically relevant and energy-matched waveforms with pulse widths from 1 to 100 s. Inter-burst broadband electrical impedance spectroscopy was performed using a low-voltage diagnostic waveform to quantify burst-resolved impedance changes. Lesions were visualized using metabolic staining, then finite element models incorporating nonlinear electroporation-dependent conductivity were used to compare anisotropic and homogenized electric field distributions. Lethal electric field thresholds were estimated by fitting simulated contours to measured lesion areas and validated using uniform electric fields generated by a parallel electrode array. Results: Across all waveforms, impedance measurements showed a rapid initial decrease followed by stabilization, indicating early electroporation saturation. Burst-to-burst percent change in impedance slope provided a consistent, waveform-agnostic metric of electroporation progression. Lesion morphology was not systematically influenced by fiber orientation, and modeling demonstrated that electroporation-induced conductivity increases homogenized tissue anisotropy. Lethal electric field thresholds increased with decreasing pulse width, ranging from 517 {+/-} 46 V/cm (100 s) to 1405 {+/-} 55 V/cm (1 s), and were validated under uniform field conditions. Conclusion: Bioimpedance-assisted monitoring enables real-time assessment of cardiac electroporation, while electroporation-induced homogenization supports simplified modeling and standardized PFA treatment design.