Omicron-Enhanced Immunosuppressive Effects of SARS-CoV-2 ORF3a and ORF9b Accessory Proteins on Monocytic Inflammatory Response
Omicron-Enhanced Immunosuppressive Effects of SARS-CoV-2 ORF3a and ORF9b Accessory Proteins on Monocytic Inflammatory Response
Grigas, J.; Padilla-Blanco, M.; Herran, U. M.; Lopez-Ayllon, B. D.; Rius, A. d. L.; Mendoza Garcia, L.; Fernandez Rodriguez, R.; Garcia Garcia, T.; Garrido, J. J.; Pautienius, A.; Stankevicius, A.; Kucinskaite-Kodze, I.; Montoya, M.
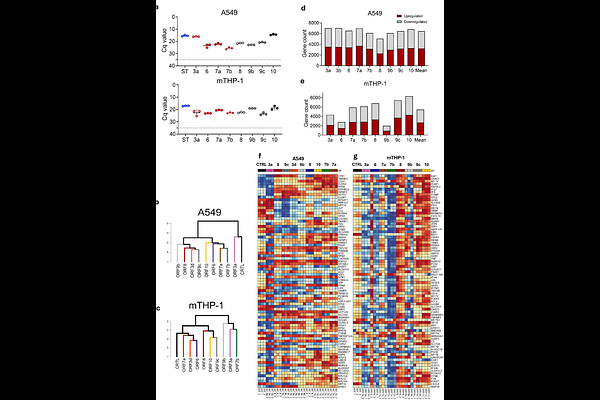
AbstractThis study investigates the poorly understood roles of SARS-CoV-2 accessory proteins using monocytic THP-1 cells expressing individual viral ORFs. ORF3a, ORF7b, and ORF9b were identified as major immunomodulators that suppress host inflammatory signaling. Specifically, cells expressing ORF3a or ORF9b exhibited reduced Toll-like receptor 4 (TLR4)-mediated production of key proinflammatory molecules - CCL2, CCL4, and IL-1 - resulting in diminished immune cell recruitment. Importantly, Omicron-associated mutations in ORF3a (T223I) and ORF9b (P10S+{Delta}E27N28A29) amplified this immunosuppressive effect, leading to stronger transcriptomic suppression consistent with Omicron's reduced pathogenicity and clinical outcomes. These findings suggest that SARS-CoV-2 accessory proteins, particularly ORF3a and ORF9b, play pivotal roles in modulating monocytic immune responses. Enhanced suppression in Omicron variants highlights an evolutionary adaptation contributing to immune evasion and milder disease manifestations.