Proteomic analysis reveals shared biological pathways linking acrolein to biomolecular changes in the acute phase of rat spinal cord injury
Proteomic analysis reveals shared biological pathways linking acrolein to biomolecular changes in the acute phase of rat spinal cord injury
Stingel, R. L.; Ball, B. K.; Sun, S.; Brubaker, D. K.; Shi, R.
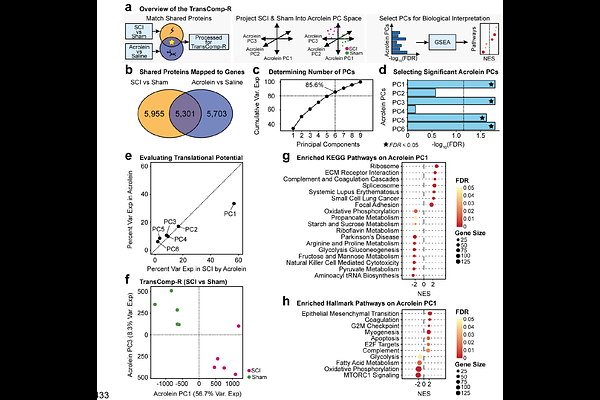
AbstractSpinal cord injury (SCI) pathology is highly difficult to treat due to substantial heterogeneity in injury presentation and spread, along with unclear mechanisms linking damage to pathology. Damages from injury forces (primary injury) are exacerbated by a series of biochemical events that follow the initial damage and injure additional tissue, known as secondary injury. Reactive aldehydes, such as acrolein, play a key role in propagating secondary injury cascades following SCI. Targeting acrolein after SCI has demonstrated therapeutic potential in limiting injury spread and pathology. However, injury mechanisms linking reactive aldehydes to SCI outcome have not been fully characterized. To gain a more comprehensive understanding of the cellular and molecular mechanisms underlying SCI, we generated proteomic profiles of rat spinal cords 24 h (acute phase) after subjection to SCI, sham injury, saline injection, or acrolein injection. We performed gene set enrichment analysis (GSEA) to characterize proteins and pathways significantly enriched after SCI and acrolein-injection. We then used Translatable Components Regression (TransComp-R), a framework for translating biological signatures across systems, to assess whether acrolein-associated spinal cord signatures can stratify SCI from sham outcomes. Our proteomics analysis revealed 467 differentially expressed proteins (DEPs) between the sham and SCI groups and 7 DEPs between saline and acrolein injection groups. Notably, the complement and coagulation cascades were upregulated in spinal cords subjected to SCI and acrolein injection. Our TransComp-R analysis further demonstrated that acrolein-associated signatures could distinguish SCI from sham conditions. Taken together, our findings suggest that acrolein induces proteomic alterations during the acute phase of SCI and is associated with complement and coagulation cascade activation, among other pathways. Therefore, this study reinforces the notion that understanding the role of acrolein in the acute phase of secondary SCI may be beneficial.