Reconstructing plant beneficial bacterial consortia by integrating dilution-to-extinction microbiome perturbation with genome-resolved synthetic ecology
Reconstructing plant beneficial bacterial consortia by integrating dilution-to-extinction microbiome perturbation with genome-resolved synthetic ecology
Jing, J.; Ossowicki, A.; Tracanna, V.; Schijlen, E.; Baak, M. L.; Pirovano, W.; van IJcken, W.; Rybka, D.; Gerards, S.; Elsayed, S. S.; Reitz, Z. L.; van Wezel, G.; Raaijmakers, J. M.; Garbeva, P.; Medema, M. H.
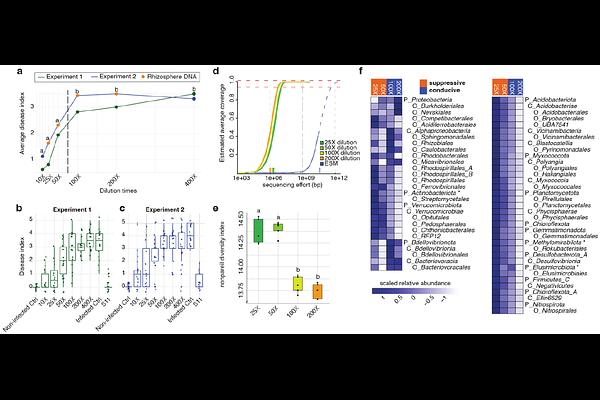
AbstractAcross the biosphere, microbiomes play essential roles in shaping the health of their host. One notable example of such a microbiome-associated phenotype is disease-suppressive soils, where susceptible plant hosts enrich and activate specific rhizosphere microbial consortia for protection against fungal root pathogens. However, identifying and reconstructing microbial consortia responsible for host protection remains challenging, given the inherent taxonomic and functional complexity of microbiomes. Here, we integrated metagenomic profiling of disease-suppressive microbiomes perturbed by dilution-to-extinction (DTE) with comprehensive culturing and synthetic ecology to identify the key bacterial taxa conferring suppressiveness to the fungal wheat pathogen Fusarium culmorum. Metagenomics of wheat rhizosphere samples along the DTE trajectory revealed bacterial taxa and functions associated with the disease-suppressive phenotype. Crosslinking these DTE metagenome data with a genome-sequenced collection of 336 rhizobacterial isolates from the suppressive soil allowed the reconstruction of synthetic communities (SynComs) of 11 de-replicated strains negatively associated with disease severity. Upon re-introduction in sterilized suppressive soils, this SynCom consistently reproduced the disease-suppressive phenotype. Paired time-series metagenomics and metatranscriptomics of the SynComs pinpointed candidate biosynthetic gene clusters, including a novel non-alpha poly-amino-acid (NAPAA) gene cluster from Arthrobacter, upregulated in presence of F. culmorum. Chemically synthesized NAPAA variants [E]-poly-L-lysine and {delta}-poly-L-ornithine significantly inhibited F. culmorum hyphal growth. Collectively, our work establishes a transformative strategy for reconstructing microbial consortia that recapitulates beneficial microbiome-associated phenotypes in plant and animal kingdoms.