Leveraging a synthetic biology approach to enhance BCG-mediated expansion of Vγ9Vδ2 T cells
Leveraging a synthetic biology approach to enhance BCG-mediated expansion of Vγ9Vδ2 T cells
Qabar, C. M.; Waldburger, L.; Keasling, J. D.; Portnoy, D. A. M.; Cox, J. S.
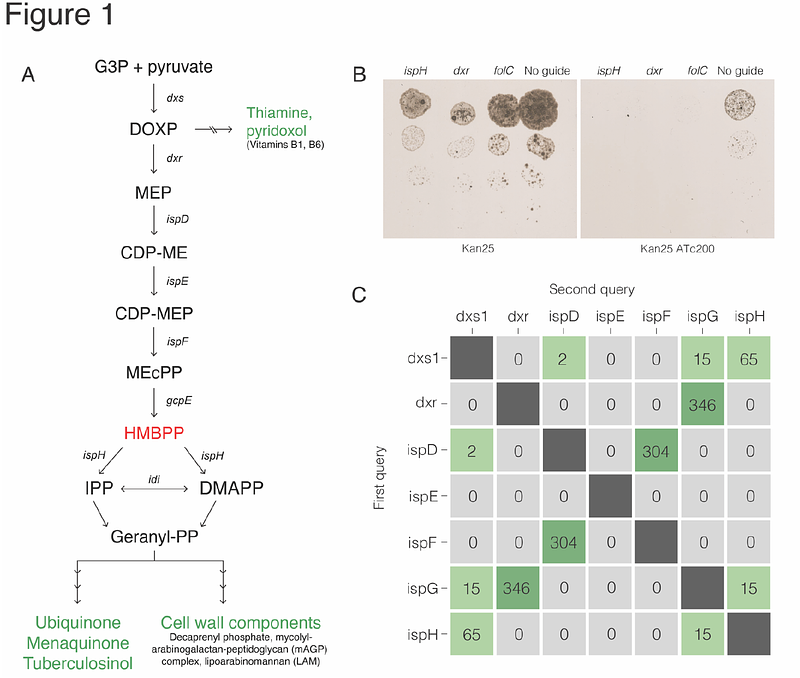
AbstractThere is an urgent need to develop a more efficacious anti-tuberculosis vaccine as the current live-attenuated vaccine strain BCG fails to prevent pulmonary infection in adults. Our long-term goal is to test whether increasing the immunogenicity of BCG will improve vaccine effectiveness while maintaining its proven safety profile. In this study, we leverage a synthetic biology approach to engineer BCG to produce more (E)-4-hydroxy-3-methyl-but-2-enyl pyrophosphate (HMBPP), a phosphoantigen produced as an intermediate of bacterial--but not host--isoprenoid biosynthesis via the methylerythritol phosphate (MEP) pathway. Importantly, HMBPP strongly activates and expands V{gamma}9V{delta}2 T cells, which are unique to higher-order primates and protect against Mycobacterium tuberculosis infection. Prior work to engineer BCG to produce specific ligands and antigens has been attempted to some success; however, our strategy exploits a self-nonself recognition mechanism in the host via HMBPP sensing, which has not been attempted before in this way. To inform the design of our recombinant strains, we performed synteny analyses of >63 mycobacterial species, which revealed that isoprenoid biosynthetic genes are not found in gene clusters or operons across all the 356 surveyed genomes. This analysis also revealed pair biases of isoprenoid biosynthesis genes frequently found in close proximity. In our engineering attempts, we found that simply overexpressing the rate-limiting gene in the pathway was toxic to the bacterium. Thus, we generated synthetic loci with the goal of specifically overproducing HMBPP, and tested the ability of these engineered strains to induce human V{gamma}9V{delta}2 T cell expansion in an in vitro stimulation assay. We found that BCG expressing a rationally-designed, synthetic MEP locus did not enhance V{gamma}9V{delta}2 T cell expansion over the wild-type vaccine strain, suggesting that ectopic expression of multiple MEP genes may result in feedback inhibition of the pathway. However we found that overexpression of the HMBPP synthase GcpE alone potently induced V{gamma}9V{delta}2 T cell expansion and did not result in downregulation of other pathway genes, presenting a successful strategy to accumulate HMBPP and overcome feedback inhibition in this pathway. While much remains to be done to ultimately develop a more efficacious vaccine, our data present a promising system to improve upon the BCG platform. To our knowledge, this is the first work to attempt reengineering of the MEP pathway in BCG to improve vaccine efficacy.