Tmn blocks phage spread via plasmolysis and triggers synergistic defence responses
Tmn blocks phage spread via plasmolysis and triggers synergistic defence responses
Wu, Y.; Zhang, Z.; Garushyants, S. K.; Li, R.; Doherty, R.; Milton, J. A.; Cooper, M. J.; Gencay, Y. E.; Amen, T.; Bakshi, S.; Patel, D. J.; Koonin, E. V.; Nobrega, F. L.
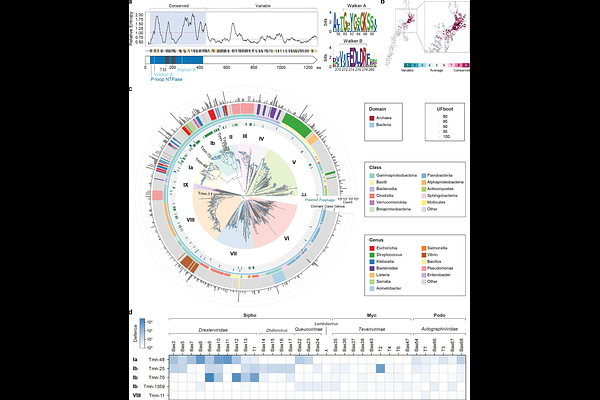
AbstractMembrane-associated phage defences remain poorly understood. Here we characterise Tmn, a YobI-family transmembrane P-loop NTPase that protects bacteria from phage infection by establishing a plasmolysis-associated antiviral state. Upon recognising phage T2 RIIB protein, Tmn enhances ATP turnover and selectively exports Mg2+, causing rapid cytoplasmic collapse that arrests phage replication without detectable membrane depolarisation or gross leakage of the cell content. Cryo-electron microscopy shows that Tmn assembles into a decameric membrane complex with extended cytosolic arms, an uncommon architecture among P-loop NTPases. The cytosolic arms, including a solenoid-like repeat domain, mediate trigger interaction and determine specificity. In addition to its primary defence function, Tmn-driven ATP collapse activates otherwise silent ATP-depletion-sensing defences, including Gabija and Septu type I, providing a mechanistic basis for synergy among defence systems and limiting secondary phage spread. These findings demonstrate that Tmn is a membrane-integrated sensor-effector that couples phage recognition to metabolic collapse and coordinated multi-layered immunity.