Cell-autonomous Wnt activity promotes transient re-programming and cell cycle re-entry of coronary artery endothelial cells
Cell-autonomous Wnt activity promotes transient re-programming and cell cycle re-entry of coronary artery endothelial cells
Bishnoi, B.; Saini, A. N.; Rao, V.; Golatkar, O.; Zirmire, R. K.; Viswanath, S.; Dhandapany, P. S.; Das, S.
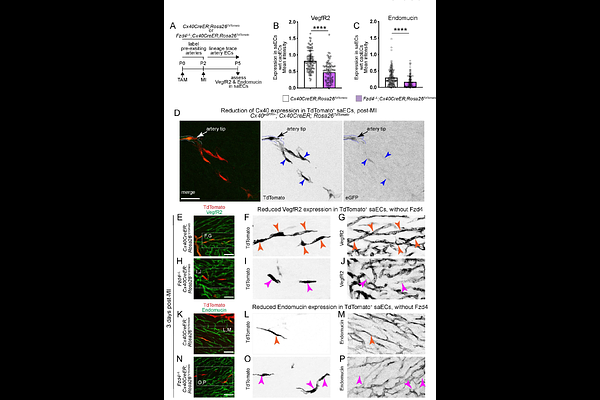
AbstractCoronary occlusion leads to formation of collateral arteries connecting occluded and perfused arteries. This process, namely artery reassembly, is temporally restricted to the cardiac regenerative window in mice and relies on cell cycle re-entry of post mitotic artery endothelial cells (ECs). Analyses of single cell RNA sequencing data of ischemic mouse hearts led to identification of Wnt activity in proliferating artery cells. Using mouse genetics and whole organ imaging at cellular resolution, we show that Wntless (Wls)-mediated Wnt secretion and Frizzled (Fzd)4-dependent Wnt reception by artery cells, was essential for development of coronary collaterals. Specifically, Fzd4 promoted proliferation and expression of (progenitor-like) capillary/venous markers in coronary artery ECs. In embryonic hearts, coronary arteries originate from capillaries. Fzd4 in these capillaries was essential for proliferation; depletion of which, accelerated their differentiation into arteries by arterialization. Along these lines, an exogeneous dose of human WNT2-sFRP1 ligand complex reactivated artery cell proliferation and restored artery reassembly in non-regenerative mouse hearts. Thus, ischemia-induced cell-autonomous Wnt signaling promoted developmental reprogramming of coronary artery ECs. Finally, exome sequencing of coronary artery disease patients, revealed unique pathogenic variants of WLS and FZD4. Together, our study provides mechanistic insights into EC plasticity and underscores the significance of WNT pathway in cardiovascular disease.