HEPLISAV-B Breaks Immune Tolerance and Induces HBV Control via CD4 T Cell-Dependent Mechanisms in a Chronic Hepatitis B Mouse Model
HEPLISAV-B Breaks Immune Tolerance and Induces HBV Control via CD4 T Cell-Dependent Mechanisms in a Chronic Hepatitis B Mouse Model
Ahodantin, J.; Wu, J.; Funaki, M.; Tang, L.; Kottilil, S.; Su, L.
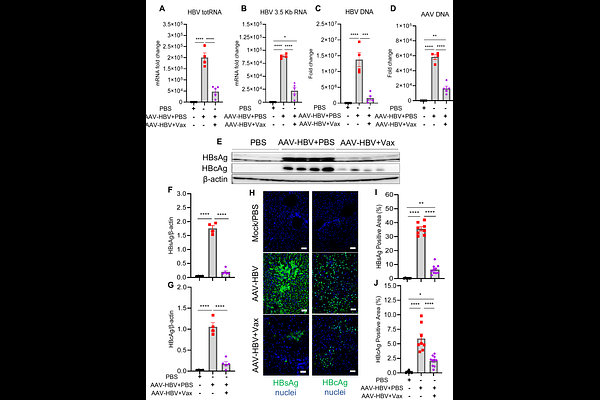
AbstractBackground: Chronic hepatitis B virus (HBV) infection (CHB) affects nearly 300 million individuals globally and remains incurable with current antiviral therapies, which suppress viral replication but rarely achieve functional cure defined by sustained loss of hepatitis B surface antigen (HBsAg). CHB is characterized by profound virus-induced immune tolerance that limits the efficacy of conventional therapeutic vaccination strategies. Objective: To evaluate the therapeutic efficacy and immunological mechanisms of HEPLISAV B, a CpG 1018 adjuvanted HBsAg vaccine, in breaking immune tolerance and inducing functional cure like responses in a murine model of CHB. Design: Using the adeno associated virus HBV (AAV HBV) mouse model, mice with high levels of persistent HBV viremia were vaccinated with two doses of HEPLISAV B. Virological outcomes in the blood and liver, immune responses and mechanisms were assessed. Results: HEPLISAV B induced rapid and durable HBsAg clearance, markedly reduced circulating and intrahepatic HBV DNA and RNA, and suppressed viral replication without hepatocellular injury. Vaccination elicited robust, sustained anti HBs IgG1 and IgA responses, enhanced HBsAg specific T and B cell immunity, reduced CD4 regulatory T cells, and decreased PD 1 expression on CD4 T cells. Therapeutic efficacy was strictly dependent on CD4 T cells and the CD40/CD40L signaling pathway, but independent of CD8 T cells, indicating a CD4 driven, non cytolytic antiviral mechanism critical for HEPLISAV B induced HBV control. Conclusion: HEPLISAV B effectively breaks HBV induced immune tolerance and restores coordinated antiviral immunity through a CD4 T cell/CD40L dependent pathway. The findings support its potential as a therapeutic vaccine in CHB patients.