TNAP and PHOSPHO1 function synergistically to afford critical control over the mineralisation of the postnatal murine skeleton
TNAP and PHOSPHO1 function synergistically to afford critical control over the mineralisation of the postnatal murine skeleton
Bourne, L. E.; Sharma, A.; Dillon, S.; Keen, J.; Jayash, S.; Crump, N.; Evans, L. A.; Karmali, M.; Promruk, W.; Clarkin, C. E.; Narisawa, S.; Stephen, L.; Foster, B. L.; Millan, J. L.; Farquharson, C.; Staines, K. A.
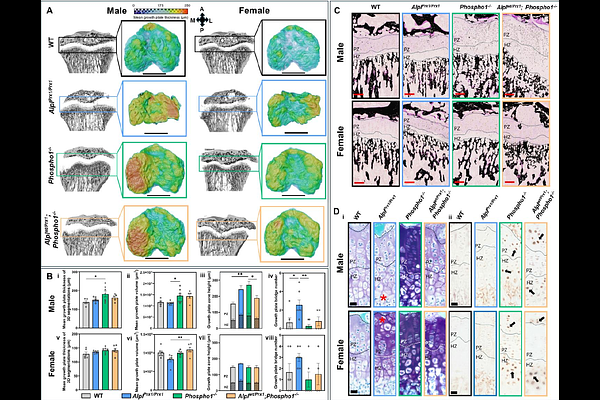
AbstractBiomineralisation is essential for skeletal integrity, yet the synergistic roles of tissue non-specific alkaline phosphatase (TNAP) and PHOSPHO1 in postnatal bone mineralisation remain poorly defined. To decipher this, we generated a novel murine model in which Alpl was deleted in Prx1-expressing cells (AlplPrx1/Prx1) in mice with a global Phospho1-/- deficiency to overcome the perinatal lethality that arises upon dual global deletion. Using a multi-modal approach to spatially phenotype the limbs of these animals, we reveal mice lacking both TNAP and PHOSPHO1 exhibit a distinct lack of mineralisation and altered anatomical structure at postnatal day 1 (PN1) and 3-weeks of age. Although viable, these mice did not thrive due to their reduced size, thus further investigations were conducted on mice with a heterozygous deletion of TNAP (Alplwt/Prx1;Phospho1-/-). Although smaller than wild-types at PN1 and 3 weeks old, these mice did not display the gross limb deformations observed in the homozygous animals and the single, functioning Alpl allele rescued the loss of biomineralisation observed following dual phosphatase deletion. At 6-weeks of age, compromised epiphyses and metaphyses were only seen in AlplPrx1/Prx1 animals. Further, we found that tibial geometry and porosity was significantly altered by Phospho1 deletion (Phospho1-/-), which was compounded in the Alplwt/Prx1;Phospho1-/- mice and linked to alterations in collagen configuration, matrix mineralisation and growth plate deformities. Together, our findings establish the mechanistic framework for TNAP and PHOSPHO1 in permissive biomineralisation, providing critical insights into this fundamental process.