Arousal as a universal embedding for spatiotemporal brain dynamics
Arousal as a universal embedding for spatiotemporal brain dynamics
Raut, R. V.; Rosenthal, Z. P.; Wang, X.; Miao, H.; Zhang, Z.; Lee, J.-M.; Raichle, M. E.; Bauer, A. Q.; Brunton, S. L.; Brunton, B. W.; Kutz, J. N.
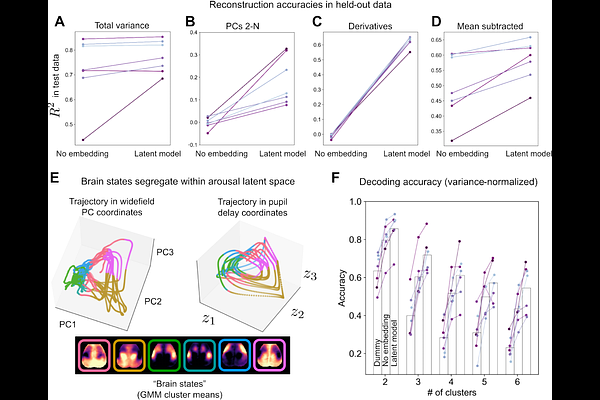
AbstractNeural activity in awake organisms shows widespread and spatiotemporally diverse correlations with behavioral and physiological measurements. We propose that this covariation reflects in part the dynamics of a unified, arousal-related process that regulates brain-wide physiology on the timescale of seconds. This interpretation motivates a surprising prediction: that a single, scalar measurement of arousal (e.g., pupil diameter) should suffice to reconstruct the evolution of multimodal, spatiotemporal measurements of large-scale brain physiology. To test this hypothesis, we perform multimodal, cortex-wide optical imaging and behavioral monitoring in awake mice. We demonstrate that spatiotemporal measurements of neuronal calcium, metabolism, and blood-oxygen can be accurately and parsimoniously modeled from a low-dimensional state-space reconstruction based on the time history of pupil diameter. Extending this framework to behavioral and electrophysiological measurements from the Allen Brain Observatory, we demonstrate the ability to integrate diverse experimental data into a unified generative model via mappings from an intrinsic arousal manifold. Our results support the hypothesis that spontaneous, spatially structured fluctuations in brain-wide physiology - widely interpreted to reflect regionally-specific neural communication - are in large part reflections of an arousal-related process. This enriched view of arousal dynamics has broad implications for interpreting observations of brain, body, and behavior as measured across modalities, contexts, and scales.