Glutamine Tautomerization Drives RhoGAP-Aided GTP Hydrolysis in Small Rho GTPases
Glutamine Tautomerization Drives RhoGAP-Aided GTP Hydrolysis in Small Rho GTPases
Parise, A.; Rozza, R.; Mitusinska, K.; Magistrato, A.
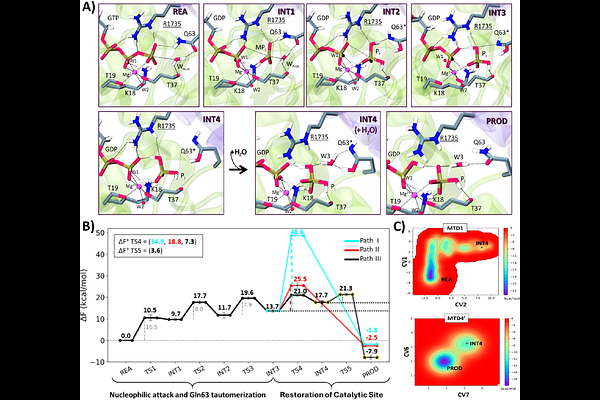
AbstractRho GTPases promote GTP hydrolysis aided by specific GTPase-activating proteins (GAPs). By alternating between an active GTP-bound and an inactive GDP-bound state, Rho GTPases function as molecular switches regulating cytoskeletal dynamics and cell motility. Despite their biological relevance, the detailed molecular mechanism underlying Rho GTPases catalysis remains contentious. Here, using classical and hybrid quantum-classical molecular dynamics, we resolve the mechanism of GTP hydrolysis in the RhoGAP-RhoA complex. We reveal that GTP hydrolysis proceeds through a dissociative nucleophilic substitution mechanism, driven by an amide [->] imide tautomerization of Gln63, which aids in delivering a proton from the nucleophilic water to the leaving phosphate group. The Gln63 imide tautomer also loosens RhoGAP-RhoA interfacial contacts, allowing solvent molecules to enter and drive a water-mediated reverse tautomerization of Gln63 that restores the catalytically-competent configuration of the RhoA active site. Conservation of key interface residues across Rho/Rho GAP family members suggests that this mechanism may be shared by most Rho GTPases.