Division state reveals hidden genetic regulation during T cell activation and identifies immune disease-linked gene programmes
Division state reveals hidden genetic regulation during T cell activation and identifies immune disease-linked gene programmes
Gozzard, M.; Sonehara, K.; Ly, K.; Rupall, T.; Ke, Z.; Ibarra-Soria, X.; Lotfollahi, M.; Soskic, B.; Jones, C. P.; Bakker, O. B.; Trynka, G.
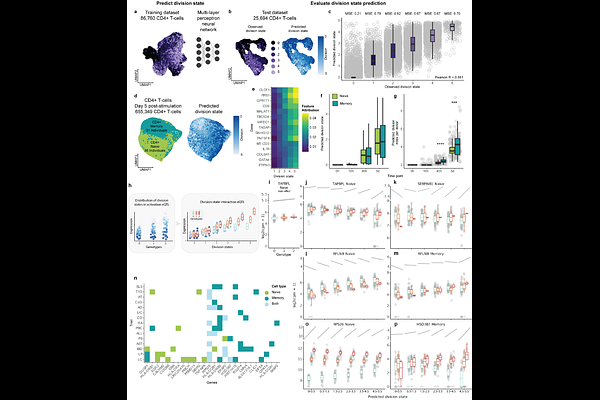
AbstractA major challenge in human genetics is to connect disease-associated variants to the cellular functions they perturb. Variants associated with immune-mediated disease have implicated CD4 T-cell activation as a disease-causal process; however, it remains unresolved whether these variants act on pathways initiating cell division, driving effector differentiation, or maintaining proliferative capacity. Activated T cells comprise mixtures of cells that have undergone different numbers of divisions, potentially obscuring genetic effects that act at specific stages of this process. Here, we study CD4 T-cell activation through cell division using division-resolved single-cell transcriptomics of naive, memory and regulatory T cells. We show that division state constitutes an important axis of transcriptional organisation, with approximately one third of genes exhibiting division-dependent regulation across shared and subset-specific programmes. We develop CellDivider, a machine-learning model that accurately infers proliferative state directly from single-cell transcriptomes, enabling a scalable approach for cohort level mapping of variant effects on cell proliferation phenotypes. Incorporating inferred division state into single-cell eQTL models reveals regulatory effects that are attenuated or missed when cells are analysed by activation time alone. Finally, we resolve gene expression programmes that govern distinct stages of T cell activation and proliferation. We show that polygenic immune disease risk preferentially converges on early activation and differentiation modules, rather than on proliferative capacity itself. Consistent with this model, division-resolved analysis of the pleiotropic immune risk gene RPS26 localises its genetic effects to early division states, providing mechanistic insight into its broad disease associations. Together, these findings illustrate how resolving dynamic cellular consequences of activation sharpens variant-to-function inference in immune disease genetics.