Imbalance of Ciliary Programs Drives Fibroblast Differentiation and Fibrotic Signaling in Systemic Sclerosis
Imbalance of Ciliary Programs Drives Fibroblast Differentiation and Fibrotic Signaling in Systemic Sclerosis
Nguyen, L. M. T.; Cordova-Fletes, C.; Sapao, P.; Vasquez-Hernandez, C.; Klumpp, A.; Nguyen, L. P. D.; Dey, P.; Gudjonsson, J. E.; Ross, R. L.; Del Galdo, F.; Riobo-Del Galdo, N. A.; Verma, P.; Varga, J.; TEVES, M. E.
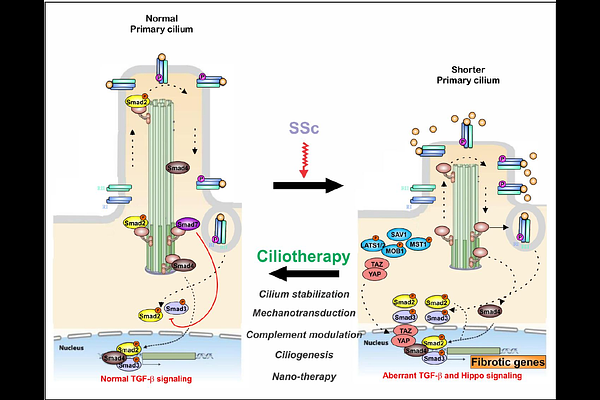
AbstractSystemic sclerosis (SSc) is a chronic fibrotic disease characterized by accumulation of activated profibrotic myofibroblasts in multiple organs. The mechanisms triggering pathogenic fibroblast to myofibroblast reprogramming in SSc, and maintaining the activated myofibroblast state, are not well known. Recent studies show that primary cilia (PC), solitary sensory organelles that integrate diverse chemical and mechanical signaling pathways, may regulate fibroblast fates. Here, using an orthogonal multimodal strategy spanning human cohorts, single-cell trajectories, and targeted perturbation models, we identify a fundamental imbalance in cilia-program dynamics as a unifying driver of fibrotic activation in SSc. Meta-analysis of skin biopsies integrating eight microarray datasets and two independent scRNA-seq cohorts, revealed a conserved 15-gene cilia signature that is altered in SSc. Single-cell trajectory mapping resolved a principal progenitor[->] secretory-like [->] myofibroblast differentiation axis, where SSc fibroblasts displayed spatially dysregulated ciliary programs that established a prolonged, disassembly-dominant "cilia-off" state. Notably, this ciliary imbalance appeared to emerge before the transition to fibrotic gene programs, positioning cilia disruption as an initiating, not secondary, event in fibroblast programming. Consistent with these transcriptomic findings, PC length was significantly reduced in SSc skin and cultured fibroblasts. Mechanistically, genetic and pharmacological perturbation studies demonstrated that cilia disruption is sufficient to drive fibrotic programs. These data establish a reciprocal regulatory framework in which shortened cilia promote sustained activation of TGF-{beta}-Hippo feed-forward signaling, driving fibroblast-to-myofibroblast transition and amplifying fibrotic responses. Together, these findings position the imbalance between ciliary assembly and disassembly as a central determinant of fibrotic fibroblast fate in SSc. Moreover, they indicate that therapeutically stabilizing primary cilia, "ciliotherapy", can reset pathological fibroblast trajectories and represents a promising antifibrotic strategy for SSc and other fibrotic conditions.