Molecular Mechanisms Governing Peptide Nanodisc Assembly and Stability
Molecular Mechanisms Governing Peptide Nanodisc Assembly and Stability
Sahoo, B. R.; Krishnarjuna, B.; Ravula, T.; Anantharamaiah, G. M.; Ramamoorthy, A.
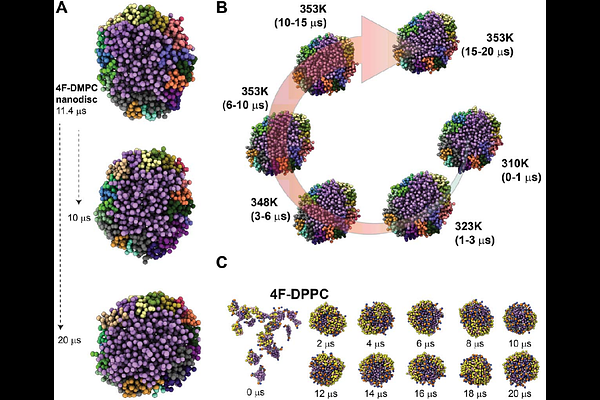
AbstractApolipoprotein A-I mimetic 4F, an 18-residue amphipathic -helix, can self-assemble with lipids to form peptide nanodiscs, yet the molecular determinants governing their assembly and stability remain poorly understood. Here, using coarse-grained molecular dynamics (CG-MD), we capture the de novo formation of 4F nanodiscs with DMPC and reveal a multistep assembly pathway involving nucleation, fusion, and ellipse-to-disc maturation. All-atom back-mapping shows that the nanodisc rim is structurally heterogeneous and stabilized by aromatic-acyl interactions, Lys/Arg headgroup anchoring, and inter-peptide electrostatic contacts. Lipid composition and temperature critically regulate nanodisc integrity: DMPC supports continuous peptide belts and long-term stability, whereas DPPC below its main phase transition temperature suppresses fusion and yields fragmented, non-uniform rims. These findings validate the ability of CG-MD to resolve nanodisc assembly mechanisms. Experimental measurements corroborate the simulations, demonstrating that 4F nanodiscs exhibit lower thermal resilience than MSP nanodiscs while retaining structural integrity at moderate temperatures. As a functional benchmark, MSP nanodiscs inhibit amyloidogenic A{beta} fibrillization, consistent with our earlier findings for 4F nanodiscs, indicating that a peptide-rimmed discoidal architecture is sufficient to suppress amyloid nucleation. Together, these results establish a mechanistic framework and design principles for single-helix peptide nanodiscs and delineate the conditions under which they converge with or diverge from MSP-based scaffolds.