High-Throughput Single-Cell Spectroscopy Using Phasor Analysis of Spectral Flow Cytometry
High-Throughput Single-Cell Spectroscopy Using Phasor Analysis of Spectral Flow Cytometry
Pannunzio, B.; Cespedes, P.; Diaz, M.; Ali, D.; Rial, A.; Malacrida, L. S.
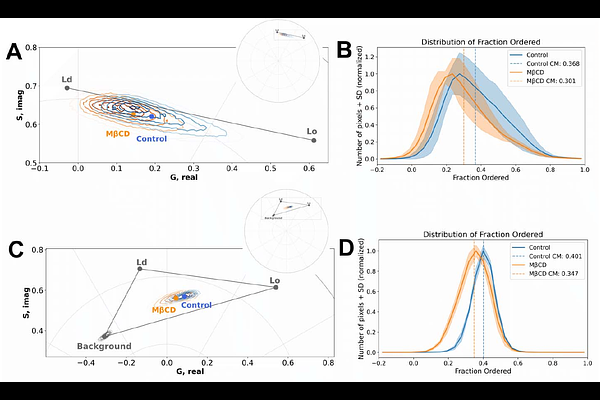
AbstractPhasor analysis is a well-established tool in hyperspectral and lifetime microscopy, providing a powerful, fit-free approach for interpreting complex fluorescence. However, its application has remained largely restricted to imaging-based modalities. Spectral flow cytometry (SFC) enables acquisition of full emission spectra from large numbers of independent single-cell events, offering superior statistical power compared to microscopy, albeit at the expense of spatial and temporal information. Here, we present the first implementation of spectral phasor analysis for SFC (phSFC), establishing a unified analytical framework that preserves interpretative continuity with hyperspectral microscopy while extending phasor-based analysis to high-throughput, single-cell measurements. Using the membrane-sensitive probe LAURDAN as a benchmark, we demonstrate that SFC reproduces phasor signatures of membrane order previously reported by hyperspectral confocal microscopy (HSI). We performed comparative analyses using multilamellar lipid vesicles (MLVs) prepared from known physical order compositions. Both modalities, SFC and HSI, accurately resolved MLVs with fluid, gel and liquid-ordered and liquid-disorder membrane phases, capturing cholesterol-dependent spectral shifts, including trajectories associated with mixtures of the different lipid phase behavior. Although absolute phasor coordinates differed between modalities due to distinct spectral sampling and detector configurations, the relative organization of membrane physical states was preserved. Notably, SFC produced more compact phasor distributions, consistent with larger sample size and enhanced statistical robustness. To further extend phSFC, we first evaluated its capacity to resolve membrane changes in live cultured cells following cholesterol depletion, establishing consistency between HSI and SFC measurements. We then applied phSFC to detect membrane dynamics in primary leukocytes isolated from bronchoalveolar lavage of mice with inflammation-associated lung pathology. LAURDAN fluorescence in the presence of autofluorescence and antibody-derived signals is quantified and discussed with simple solution by n-harmonic phasor analysis unmixing. Together, these results establish SFC as a robust and complementary extension of LAURDAN phasor analysis, bridging HSI and high-throughput flow cytometry measurements.