Axial Nephron Fate Switching Demonstrates a Plastic System Tunable on Demand
Axial Nephron Fate Switching Demonstrates a Plastic System Tunable on Demand
Achieng, M. A.; Schnell, J.; Fausto, C. C.; Csipan, R. L.; Thornton, M. E.; Grubbs, B. H.; Lindström, N. O.
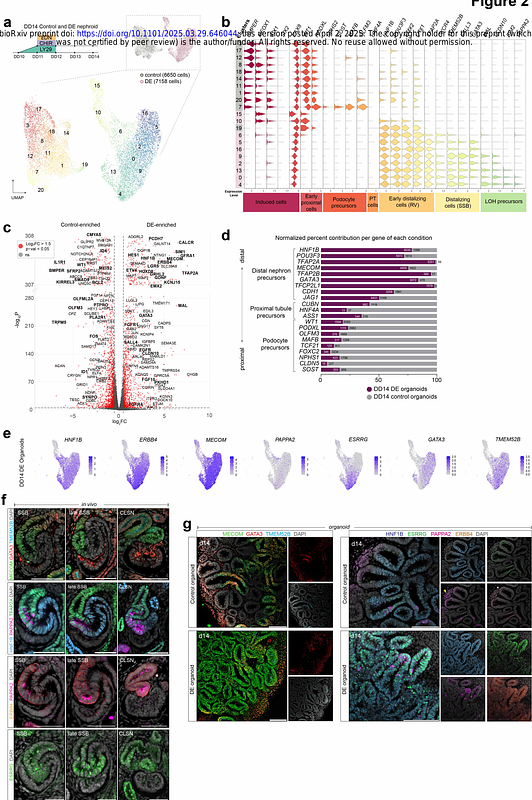
AbstractThe human nephron is a highly patterned tubular structure. It develops specialized cells that regulate bodily fluid homeostasis, blood pressure, and urine secretion throughout life. Approximately 1 million nephrons form in each kidney during embryonic and fetal development, but how they develop is poorly understood. Here we interrogate axial patterning mechanisms in the human nephron using an iPSC-derived kidney organoid system that generates hundreds of developmentally synchronized nephrons, and we compare it to in vivo human kidney development using single cell and spatial transcriptomic approaches. We show that human nephron patterning is controlled by integrated WNT/BMP/FGF signaling. Imposing a WNTON/BMPOFF state established a distal nephron identity that matures into thick ascending loop of Henle cells by endogenously activating FGF. Simultaneous suppression of FGF signaling switches cells back to a proximal cell-state, a transformation that is in itself dependent on BMP signal transduction. Our system highlights plasticity in axial nephron patterning, delineates the roles of WNT, FGF, and BMP mediated mechanisms controlling nephron patterning, and paves the way for generating nephron cells on demand.