A sensor of oxidative stress confers virulence via response memory in Acinetobacter baumannii
A sensor of oxidative stress confers virulence via response memory in Acinetobacter baumannii
Ngo, H. V.; Kim, S. H.; Ha, H.; Kang, S.; Shin, D.; Gunzer, M.; Kim, K.; Yeom, J.
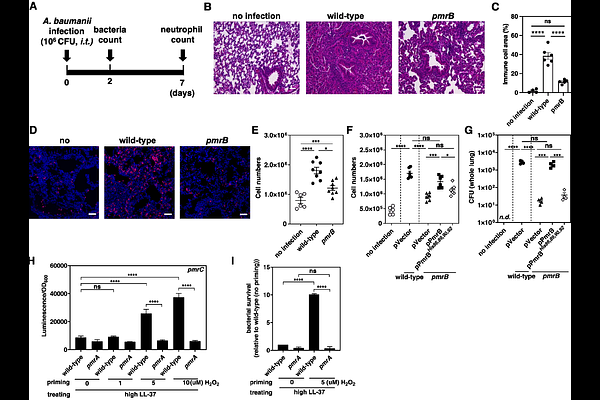
AbstractAll living organisms must adapt to environmental stresses to survive. The two-component system (TCS) is a prevalent signal transduction mechanism to alter gene expression in response to stress in bacteria. We report that the PmrB sensor of the PmrA/PmrB TCS senses sublethal oxidative stress and encodes a response memory that promotes virulence in Acinetobacter baumannii. PmrB detects oxidative stress through histidine residues with a nickel (Ni2+) cofactor. Nickel oxidation (Ni2+) induces an allosteric effect by altering PmrB conformation, enabling PmrA activation. Activated PmrA induces antioxidant defense genes, including iron-sulfur cluster repair, ferritin, catalase, and peroxidase. Notably, PmrB remains activated after sublethal oxidative stress is removed, as pathogens encounter in the bloodstream and airways during infection. This response memory enables bacteria to respond strongly to subsequent lethal oxidative stress and antimicrobial peptides. In a murine model, disrupting oxidative sensing reduces virulence. In addition, PmrB-mediated response memory is essential for high virulence in clinically isolated multidrug-resistant A. baumannii. Our findings demonstrate how subinhibitory stress detection enables pathogens to achieve full virulence potential and resist stresses during infection.