Stage-specific metabolic rewiring coordinates nucleotide supply and demand during spermatogenesis
Stage-specific metabolic rewiring coordinates nucleotide supply and demand during spermatogenesis
Paz, G. B.; Mayorek, N.; Mansour, Y.; Stein, I.; Medvedev, E.; Elgavish, S.; Ruppo, S.; Vardi, O.; Sarvin, B.; Sarvin, N.; Shlomi, T.; Pikarsky, E.
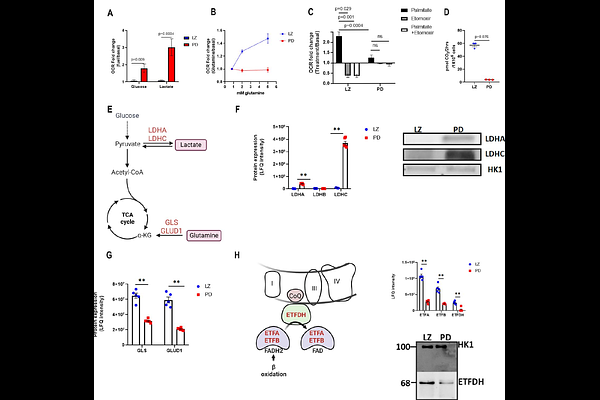
AbstractMale meiotic prophase I features a major transition from leptotene/zygotene (LZ) to pachytene/diplotene (PD) spermatocytes, accompanied by rapid growth and extensive transcriptional remodeling. Using stage-resolved purification of spermatogenic populations integrated with Seahorse respirometry, proteomics/metabolomics, and stable-isotope tracing, we define metabolic programs across this transition. LZ spermatocytes show little oxidative response to glucose or lactate and instead rely on glutamine and CPT1-dependent fatty-acid {beta}-oxidation to sustain mitochondrial respiration. In contrast, PD spermatocytes increase oxygen consumption upon glucose and lactate addition and suppress fatty-acid oxidation. U-[13C]-glucose tracing reveals preferential diversion of glucose-6-phosphate into the pentose phosphate pathway (PPP) in LZ cells, whereas PD cells lose PPP entry coincident with meiotic sex chromosome inactivation and repression of the X-linked enzyme G6PD. Isotope incorporation into UTP/CTP demonstrates active de novo pyrimidine synthesis in LZ cells via PPP-derived ribose and pyruvate carboxylase/TCA-derived aspartate, while PD cells show minimal labeling. Despite silencing nucleotide biosynthesis, PD cells exhibit elevated nascent RNA synthesis and reduced nucleotide pools, consistent with increased nucleotide demand. Notably, pharmacologic inhibition of ENT1, the only nucleoside transporter expressed in these cells, abolishes in vivo 5-ethynyl uridine uptake in early germ cells yet does not impair meiotic progression. This indicates that germ cells do not depend on external nucleoside sources and that PD-stage transcriptional demands are met primarily by nucleotide pools synthesized earlier. Cross-species transcriptomics (mouse, human, zebrafish, yeast) reveals a conserved shutdown of nucleotide biosynthesis during late prophase I. Together, our results support a developmentally enforced separation between nucleotide production in early prophase and nucleotide utilization in pachytene.