A shared spatial topography links the functional connectome correlates of cocaine use disorder and dopamine D2/3 receptor densities
A shared spatial topography links the functional connectome correlates of cocaine use disorder and dopamine D2/3 receptor densities
Ricard, J. A.; Labache, L.; Segal, A.; Dhamala, E.; Cocuzza, C. V.; Jones, G.; Yip, S.; Chopra, S.; Holmes, A. J.
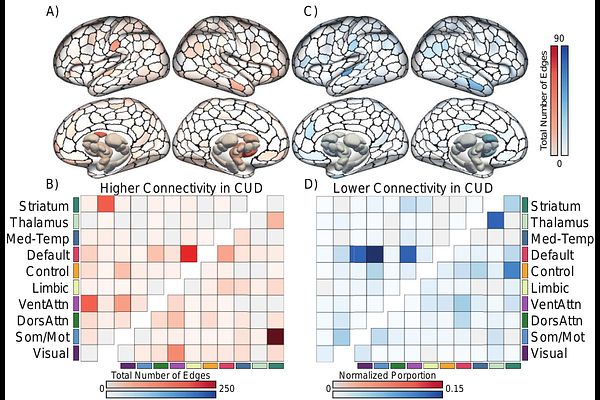
AbstractBackground: The biological mechanisms that contribute to cocaine and other substance use disorders involve an array of cortical and subcortical systems. Prior work on the development and maintenance of substance use has largely focused on cortico-striatal circuits, with relatively less attention on alterations within and across large-scale functional brain networks, and associated aspects of the dopamine system. The brain-wide pattern of temporal co-activation between distinct brain regions, referred to as the functional connectome, underpins individual differences in behavior. Critically, the functional connectome correlates of substance use and their specificity to dopamine receptor densities relative to other metabotropic receptors classes remains to be established. Methods: We comprehensively characterized brain-wide differences in functional connectivity across multiple scales, including individual connections, regions, and networks in participants with cocaine use disorder (CUD; n=69) and healthy matched controls (n=62), Further, we studied the relationship between the observed functional connectivity signatures of CUD and the spatial distribution of a broad range of normative neurotransmitter receptor and transporter bindings as assessed through 18 different normative positron emission tomography (PET) maps. Results: Our analyses identified a widespread profile of functional connectivity differences between individuals with CUD and matched healthy comparison participants (8.8% of total edges; 8,185 edges; pFWE=0.025). We largely find lower connectivity preferentially linking default network and subcortical regions, and higher within-network connectivity in the default network in participants with CUD. Furthermore, we find consistent and replicable associations between signatures of CUD and normative spatial density of dopamine D2/3 receptors. Conclusions: Our analyses revealed a widespread profile of altered connectivity in individuals with CUD that extends across the functional connectome and implicates multiple circuits. This profile is robustly coupled with normative dopamine D2/3 receptors densities. Underscoring the translational potential of connectomic approaches for the study of in vivo brain functions, CUD-linked aspects of brain function were spatially coupled to disorder relevant neurotransmitter systems.