The mitochondrial calcium uniporter is necessary for synaptic plasticity and proper mitochondrial morphology and distribution in the distal dendrites of CA2 neurons
The mitochondrial calcium uniporter is necessary for synaptic plasticity and proper mitochondrial morphology and distribution in the distal dendrites of CA2 neurons
Pannoni, K. E.; Fischer, Q. S.; Tarannum, R.; Cawley, M. L.; Alsalman, M. M.; Acosta, N.; Ezigbo, C.; Gil, D. V.; Campbell, L. A.; Farris, S.
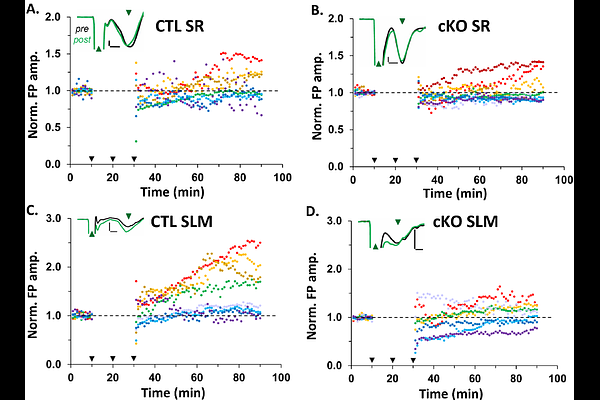
AbstractMitochondria are dynamic organelles that are morphologically and functionally diverse across different cell types and subcellular compartments in order to meet unique energy demands. In neurons, mitochondria are critical to support synapses and synaptic plasticity. However, the mechanisms regulating mitochondria in synaptic plasticity are largely unknown. The mitochondrial calcium uniporter (MCU) regulates calcium entry into the mitochondria, which in turn regulates the bioenergetics and distribution of mitochondria to active synapses. Evidence suggests that calcium influx via MCU couples neuronal activity to mitochondrial metabolism and ATP production, which would allow neurons to rapidly adapt to changing energy demands. Intriguingly, MCU is uniquely enriched in CA2 distal dendrites relative to neighboring CA1 or CA3 distal dendrites, suggesting mitochondria there are molecularly distinct. However, the functional significance of this enrichment is not clear. Synapses onto CA2 distal dendrites, unlike synapses onto CA2 proximal dendrites, readily undergo long-term potentiation (LTP), but the mechanisms underlying the different plasticity profiles are unknown. Therefore, we investigated the role of MCU in regulating dendritic mitochondria and synaptic plasticity in CA2 distal dendrites. Using a CA2-specific MCU knockout (cKO) mouse, we found that MCU is required for LTP at CA2 distal dendrite synapses. Loss of LTP correlated with a trend for decreased spine density in CA2 distal dendrites of cKO mice compared to control (CTL) mice, which was predominantly seen in immature spines Moreover, mitochondria were significantly smaller and more numerous across all dendritic layers of CA2 in cKO mice compared to CTL mice, suggesting an overall increase in mitochondrial fragmentation. Fragmented mitochondria might have functional changes, such as altered ATP production, that might explain a deficit in synaptic plasticity. Collectively, our data reveal that MCU regulates layer-specific forms of plasticity in CA2 dendrites, potentially by maintaining proper mitochondria morphology and distribution within dendrites. Differences in MCU expression across different cell types and circuits might be a general mechanism to tune the sensitivity of mitochondria to cytoplasmic calcium levels to power synaptic plasticity.