The disordered and structured regions of α-Synuclein contribute to membrane remodeling synergistically
The disordered and structured regions of α-Synuclein contribute to membrane remodeling synergistically
Johnson, D. H.; Liow, J. S.; Kou, O. H.; Zeno, W. F.
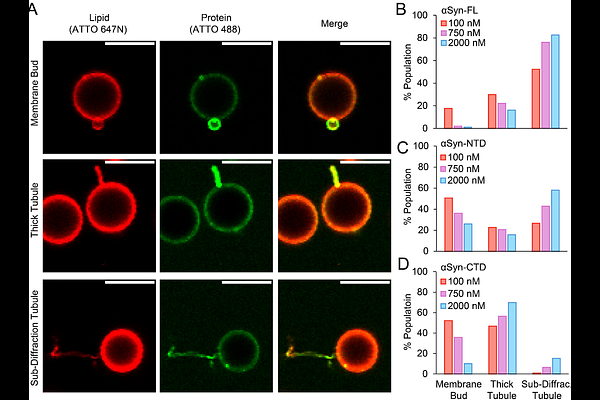
Abstract-Synuclein (Syn) remodels cellular membranes through interactions that involve both its structured, membrane-binding N-terminal domain (NTD) and intrinsically disordered C-terminal domain (CTD). While the amphipathic NTD helix is known to insert into lipid bilayers and generate curvature, the contribution of the acidic CTD remains unclear. Here, we dissect the individual and cooperative roles of these domains using Supported Bilayers with Excess Membrane Reservoir (SUPER) templates to quantify membrane remodeling via membrane fission and membrane morphological deformations (i.e., membrane budding and tubulation). We show that both the NTD and CTD independently remodel membranes, while full-length Syn exhibits greater remodeling ability than either the NTD or CTD in isolation. This result demonstrates a synergistic amplification between helix insertion of the NTD and the tethered, disordered CTD. To further probe the mechanism of membrane remodeling by the CTD, we modulated the chain length of the protein, the bulk ionic strength of the solution (i.e., charge screening), and applied relevant polymer scaling laws for disordered proteins. Our results suggest that the membrane remodeling mechanism for the disordered CTD is electrostatic in nature, stemming from protein-protein repulsion at elevated binding densities. Together, our findings reveal a cooperative energetic mechanism in which N-terminal helix insertion biases membrane curvature and the disordered, C-terminal domain adds an additional electrostatic component that helps to overcome the free energy barrier for membrane bending.