A microfluidic platform for multi-marker profiling of extracellular vesicles from single-cell-derived clones
A microfluidic platform for multi-marker profiling of extracellular vesicles from single-cell-derived clones
Kim, J.; Petrisor, D.; Stoianovici, D.; Amend, S.; Pienta, K.; Kim, C.-J.
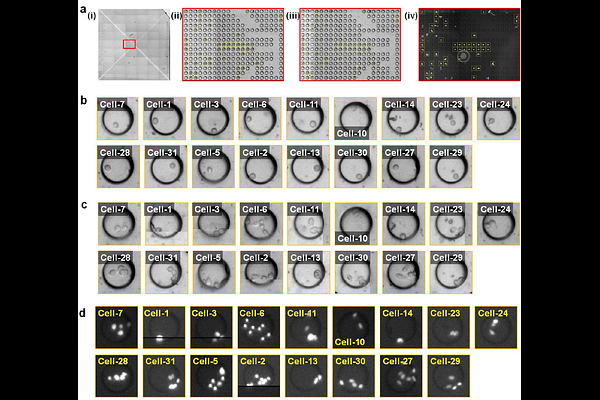
AbstractExtracellular vesicles (EVs) carry molecular cargo that can reflect the real-time state of parental cells, yet most in vitro EV analyses rely on bulk approaches and therefore average over pronounced heterogeneity in both cell and EV populations. Here, we present a semi-open microfluidic platform that enables multi-marker profiling of EVs released from single-cell-derived clones, allowing EV signatures to be linked to clonal progeny originating from a single parental cell. The platform integrates aligned cell and EV arrays containing 17,305 wells, assembled with a 3D-printed housing to capture released EVs in one-to-one matched wells. Captured EVs are immunolabeled for canonical tetraspanin markers (CD9, CD63, CD81) and EpCAM, imaged by high-resolution fluorescence microscopy, and quantified using an automated image-analysis pipeline. Applying the platform to single-cell-derived PC3 clones revealed substantial heterogeneity in EV marker co-expression, with hierarchical clustering identifying four distinct tetraspanin co-expression profiles. The fraction of EpCAM-positive EVs increased with PC3 cell proliferation, as assessed by endpoint cell number, whereas free (non-EV-associated) EpCAM showed no correlation. This platform enables near single-EV-level, multi-marker profiling from single-cell lineages and provides a practical approach to simultaneously dissect both cellular and EV heterogeneity.