Structure and mechanism of the human TMEM260 O-mannosyltransferase
Structure and mechanism of the human TMEM260 O-mannosyltransferase
Cifuente, J. O.; Povolo, L.; Ochoa-Lizarralde, B.; Ahmed, S.; Vakhrushev, S. Y.; Lopez-Alonso, J. P.; Tascon, I.; Fernandez-Martinez, J.; Joshi, H. J.; Halim, A.; Ubarretxena-Belandia, I.
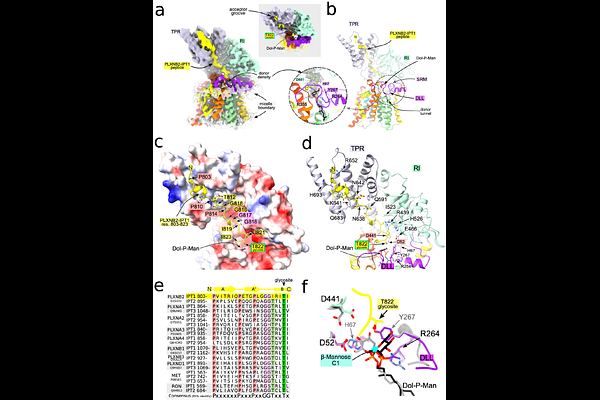
AbstractProtein O-linked mannose (O-Man) glycosylation is essential for mammalian development, and mutations in its biosynthetic glycosyltransferases cause severe muscular, neurological and cardiac disorders. Despite its biological and clinical importance, the structural basis of mammalian O-Man biosynthesis has remained unknown. Here we report cryo-electron microscopy (cryo-EM) structures of human TMEM260, an endoplasmic reticulum glycosyltransferase that selectively catalyzes O-mannosylation of semaphorin plexin receptors and receptor tyrosine kinases cMET and RON, key regulators of cell guidance and migration. Structures of TMEM260 in a ternary complex with its natural donor dolichyl-phosphate-{beta}-mannose (Dol-P-Man) and an acceptor peptide derived from plexin-B2, together with binary complexes with Dol-P-Man or a synthetic donor analogue, capture physiologically relevant, substrate-loaded states and reveal the structural basis of O-Man transfer. We identify a conserved O-mannosylation sequon that underlies acceptor specificity and show that TMEM260 modifies extended polypeptide substrates, consistent with a co-translational glycosylation mechanism. These findings establish the molecular mechanism of a mammalian O-mannosyltransferase required for the maturation of physiologically critical receptors and provide a structural framework for interpreting TMEM260-associated congenital malformations.