Extending the MARTINI 3 Coarse-Grained Forcefield to Polypeptoids
Extending the MARTINI 3 Coarse-Grained Forcefield to Polypeptoids
Wang, J.; Yu, Z.; Zhao, M.
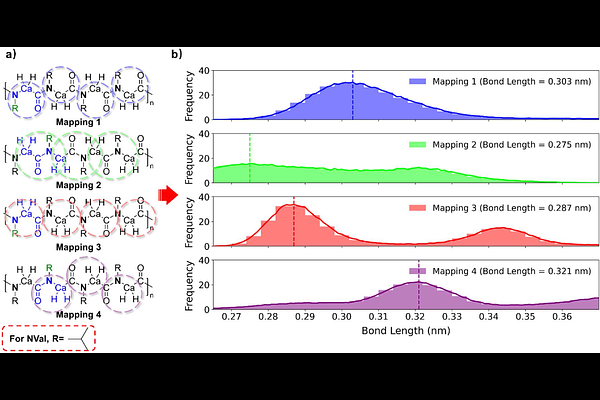
AbstractPolypeptoids (poly-N-substituted glycines) are synthetic peptidomimetic polymers with their sidechains attached to the backbone amide nitrogen rather than the -carbon in natural peptides. Peptoids display pronounced sequence-dependent conformational flexibility arising from the absence of backbone hydrogen bonding and slow cis/trans {omega}-dihedral isomerization. Despite growing interest in peptoid-based biomaterials, a coarse-grained (CG) model compatible with the modern MARTINI 3 framework is not yet available, limiting mesoscale simulation of peptoid structure and self-assembly. In this work, we develop the first MARTINI 3 compatible peptoid CG forcefield, covering 19 commonly used residue types. Extensive all-atom reference simulations employing parallel bias metadynamics (PBMetaD) were performed to ensure converged sampling of {omega}-dihedral transitions. Bonded parameters were derived from atomistic distribution functions via direct Boltzmann inversion (DBI), while nonbonded interactions were primarily adopted from the standard MARTINI 3 parameter library. The resulting CG model reproduces structural and thermodynamic properties in close agreement with all-atom simulations, while providing up to 57-fold enhanced computational efficiency. To facilitate its adoption by the research community, we have integrated all parameters and workflows to the MARTINI-based martinize2 tool, enabling automated generation of MARTINI 3 peptoid structures and topologies. This work establishes a transferable and computational efficient framework for simulating large-scale peptoid confirmations, assemblies, membrane interactions, and nanostructure formation, and supports the rational design of next-generation sequence-specific functional peptoid-based materials.