Molecular Determinants of Allosteric Inhibitor Affinity and Selectivity in PDE5
Molecular Determinants of Allosteric Inhibitor Affinity and Selectivity in PDE5
Verma, J.; Vashisth, H.
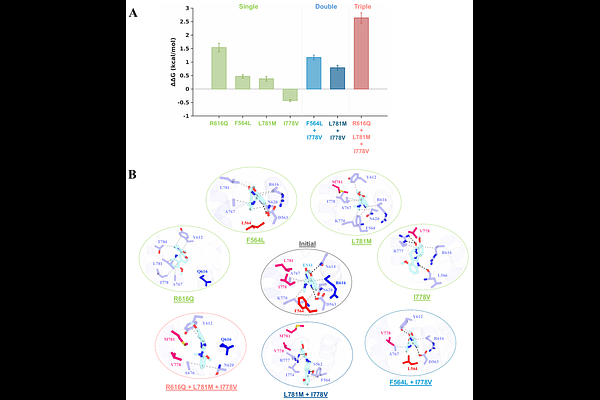
AbstractAllosteric inhibitors targeting unique regulatory pockets provide an opportunity for selective modulation of proteins within the phosphodiesterase (PDE) family. In this work, we investigate the molecular determinants for binding of an evodiamine derivative (EVO) to PDE5 and the structural basis for differential recognition of this inhibitor in PDE6 isoforms. We integrated structural modeling, all-atom molecular dynamics (MD) simulations, and extensive alchemical free-energy calculations to quantify residue-level contributions to inhibitor binding in PDE5 through systematic alanine scanning and PDE6-derived mutations. Thermodynamic analysis based on free energy identified key residues that govern EVO binding through a network of hydrogen bonds and hydrophobic contacts. Specifically, we examined structural changes associated with allosteric pocket variations in PDE5, identified key structural elements, and evaluated the role of chemical substituents of EVO in allosteric inhibition. Moreover, PDE6-derived substitutions revealed isoform specific effects, with PDE6-rod substitutions resulting in stronger destabilization than PDE6-cone substitutions. Together, these results define the energetic and structural basis for allosteric inhibition of PDE5 and provide mechanistic insights for the rational design of selective PDE inhibitors.