DM: a simple solution to suppress air-water interface interactions in cryo-EM
DM: a simple solution to suppress air-water interface interactions in cryo-EM
Rafiq, M.; Schaefer, J.-H.; Rahmani, H.; You, S.; Bollong, M. J.; Grotjahn, D.; Wiseman, L.; Lander, G. C.
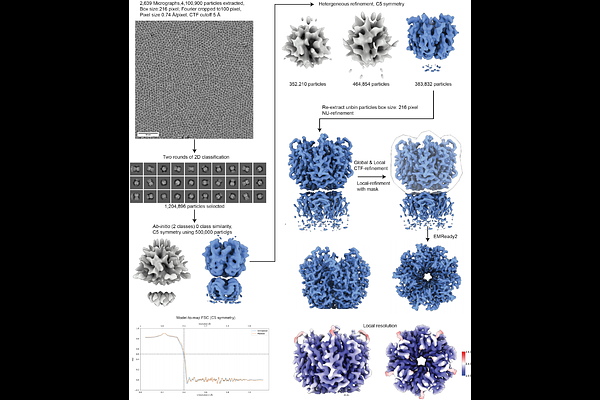
AbstractThe air-water interface remains the primary barrier to routine high-resolution cryo-EM structure determination, driving protein adsorption, structural denaturation, and restricted particle orientations during vitrification. Here, we describe a simple and broadly applicable strategy to mitigate these effects using the mild non-ionic detergent n-decyl-{beta}-D-maltopyranoside (DM). Addition of DM at low millimolar concentrations immediately prior to vitrification consistently suppresses AWI-driven artifacts, resulting in improved angular sampling, reduced structural damage, and enhanced reconstruction quality across diverse macromolecular systems. Using this approach, we obtained a high-resolution reconstruction of the 65 kDa Nucleophosmin1 pentamer, a target previously limited by severe preferred orientation issues. We further show that DM promotes isotropic particle distributions for high-resolution reconstruction of hemagglutinin, transthyretin, as well as suppressing denaturation of aldolase while stabilizing its C-terminus. Our results indicate that DM effectively passivates deleterious air-water interface interactions without compromising particle integrity. These results establish DM as an effective additive for improving the robustness of single-particle cryo-EM sample preparation.