Global genomic epidemiology of Candida auris: analysis of 12,644 whole genome sequences from 1997-2024
Global genomic epidemiology of Candida auris: analysis of 12,644 whole genome sequences from 1997-2024
Gifford, H.; Helmstetter, N.; Zerrouki, H.; Wilson, D.; Rhodes, J.; Farrer, R. A.
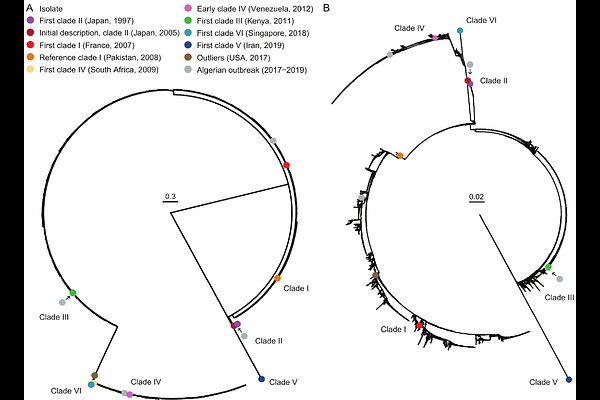
AbstractCandida auris is a critical priority fungal pathogen (World Health Organization) that has emerged into human populations pre-1996 from an unknown environmental reservoir. Genomic sequencing data has been used extensively in the last decade, leading to a largely unharnessed dataset with potential to unlock understanding of emerging fungal pathogen evolution. Here, we compiled publicly available Illumina paired-end whole genome sequences (WGS) for variant calling, supplemented by isolates from the worlds first four-clade single-facility outbreak in Algeria (n = 7), totaling 12,644 WGS. We describe the geographic, clinical, and temporal epidemiology of the outbreak across the globe between 1997 and 2024, indicating a conserved six-clade structure. Despite the evidence of earlier species diversity in low- and lower-middle income countries (LLMIC), the majority of WGS derived from high-income (12,035, 93.2%) and upper-middle-income countries (719, 5.57%), where cases are believed to be imported from endemic regions, with few from lower-middle-income countries (165, 1.28%) and none from lower-income countries. Copy number variation was present, including azole drug target ERG11, with seven isolates displaying ten to fifteen copies. Alarmingly, the standing variation of C. auris reveals emerging variation in hot-spots of the echinocandin drug target FKS1, which encodes a {beta}-1,3-glucan synthase. Eighteen emerging FKS1 hot-spot variants have not been detected or described in databases, and mostly occured in Northern America (57/61, 93.4%), where echinocandin monotherapy is standard treatment. Well-studied FKS1 variants known to cause resistance are significantly more common in clade I isolates derived from urine compared to blood, consistent with a role for the urinary niche as a low echinocandin concentration safe-haven for the development of resistance. The insights from this global genomic epidemiology survey of C. auris highlight sequencing inequality and detect ongoing genomic innovation in clinical settings, raising predictable and urgent concerns around the ongoing use of echinocandin monotherapy and potential emerging antifungal drug resistance-related genotypes in high-income settings.