Defect in hematopoiesis and embryonic lethality at midgestation of Vps13a/Vps13c double knockout mice
Defect in hematopoiesis and embryonic lethality at midgestation of Vps13a/Vps13c double knockout mice
Xu, P.; Mancuso, R. I.; Leonzino, M.; Zeiss, C. J.; Krause, D. S.; De Camilli, P.
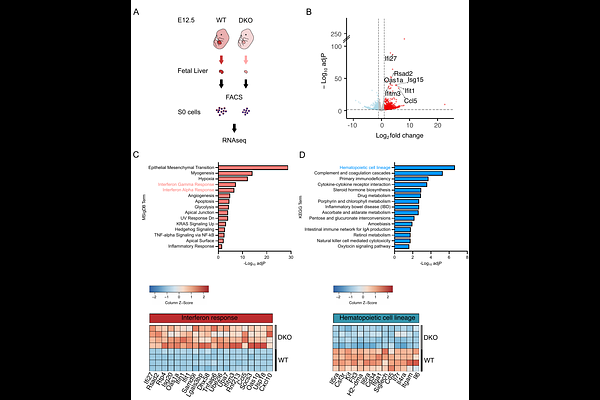
AbstractVPS13 is the founding member of a family of proteins that mediate lipid transfer at intracellular membrane contact sites by a bridge-like mechanism. Mammalian genomes comprise 4 VPS13 genes encoding proteins with distinct localizations and function. The gene duplication resulting in VPS13A and VPS13C is the most recent in evolution and, accordingly, these two proteins are the most similar to each other. However, they have distinct subcellular localizations and their loss of function mutations in humans are compatible with life but result in two different age-dependent neurodegenerative diseases, chorea-acanthocytosis and Parkinson\'s disease, respectively. Thus, it remains unclear whether these two proteins have overlapping functions. Here, we show that while Vps13a KO and Vps13c KO mice are viable, embryonic development of Vps13a/Vps13c double knockout (DKO) mice is arrested at midgestation. Prior to death, DKO embryos were smaller than controls, were anemic and had a smaller liver, the key erythropoietic site at this developmental stage. Further analyses of erythroid precursor cells showed that their differentiation was impaired and that this defect was accompanied by activation of innate immunity as revealed by upregulation of interferon stimulated genes (ISGs). Additionally, the RIG-I and MDA5 components of dsRNA triggered innate immunity were found upregulated in the DKO fetal liver. Activation of innate immunity may result from loss of integrity of the membranes of intracellular organelles, such as mitochondria and autophagic lysosomes, due to the absence of these lipid transport proteins. The surprising and striking synthetic effect resulting for the combined loss of VPS13A and VPS13C suggests that despite of the different localization of these two proteins, the lipid fluxes that they mediate are partially redundant.