Depletion of Chloroplast HSP70B Triggers Proteostasis Collapse and Compromises Thylakoid Membrane Integrity in Chlamydomonas
Depletion of Chloroplast HSP70B Triggers Proteostasis Collapse and Compromises Thylakoid Membrane Integrity in Chlamydomonas
Probst, A.; Schmollinger, S.; Berg, J.; Unger, A.-K.; Strenkert, D.; Geimer, S.; Sommer, F.; Schroda, M.
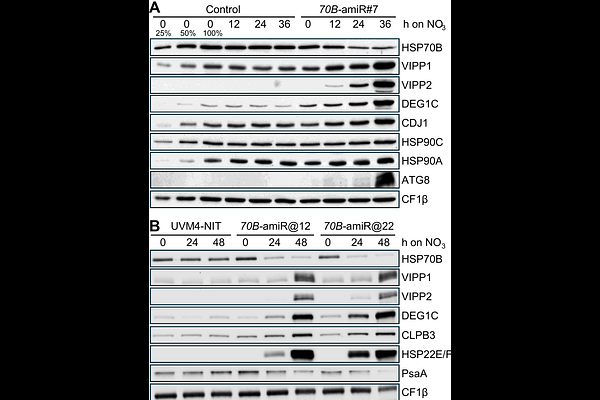
AbstractChloroplast HSP70 is an essential component of the plastid proteostasis network, supporting protein folding, complex assembly and disassembly, and stress acclimation. Despite extensive genetic evidence for its essentiality, the cellular consequences of reduced chloroplast HSP70 activity remain poorly defined. Here, we investigated the function of the sole chloroplast HSP70 in Chlamydomonas reinhardtii, HSP70B, using an inducible artificial microRNA approach that reduced HSP70B abundance to below 30% of wild-type levels. HSP70B depletion resulted in cell division arrest and extensive proteome remodeling, characterized by strong upregulation of proteins involved in chloroplast protein quality control and membrane remodeling. Notably, this response was accompanied by increased abundance of protein quality control components in the endoplasmic reticulum, cytosol, and mitochondria, indicating pronounced proteostasis cross-talk between cellular compartments. In contrast, chloroplast and cytosolic ribosomes, photosynthetic and respiratory protein complexes, and central metabolic enzymes were broadly depleted, consistent with a collapse of cellular proteostasis. At the ultrastructural level, HSP70B-depleted cells exhibited lesions at thylakoid membrane conversion zones previously described in VIPP1-depleted cells. Accordingly, higher-order oligomeric forms of VIPP1 accumulated, and cells displayed extreme sensitivity to high-light stress. These findings confirm HSP70B as a key regulator of VIPP1 oligomer dynamics and highlight its central role in coordinating chloroplast membrane remodeling with cellular proteostasis in Chlamydomonas.