Placental prostaglandin signaling disrupts barrier integrity and relays an acute inflammatory signal to the fetus.
Placental prostaglandin signaling disrupts barrier integrity and relays an acute inflammatory signal to the fetus.
Horackova, H.; Zhao, Q.; Faulkner, S.; Alvarez, J.; Akiz, C.; Liu, Y.; Crosthwait, J.; Dai, W.; Santoyo, D.; Pham-Chang, D.; Bhinderwala, N.; Tagliaferro, T.; Wallace, W.; Walia, S.; Martinovic, J.; Baldauf, C.; Montagne, A.; Bonnin, A.
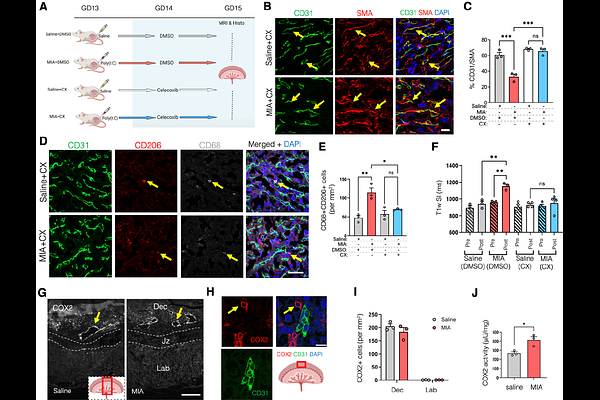
AbstractMaternal inflammation during pregnancy is a major risk factor for adverse neurodevelopmental outcomes, yet the mechanisms linking maternal immune activation (MIA) to placenta-fetal brain axis dysfunction remain unclear. Using a poly-(I:C) mouse model, we show that MIA rapidly disrupts placental-blood barrier (PBB) integrity by disrupting pericyte-endothelium coupling within 48 hours, leading to increased placental permeability detected by in vivo MRI. We identify a prostaglandin E2 (PGE2)-dependent pathway involving cyclooxygenase-2 (COX2) and prostaglandin E-synthase (mPGES1) as a key mediator of this effect. Pharmacological inhibition of COX2 prevented MIA-induced structural and functional changes. In vitro explant studies confirmed that PGE2 acts via the EP3 receptor to disrupt pericyte-endothelium interactions, while ex vivo perfusion demonstrated that in addition to its local effects on PBB structure, placenta-derived PGE2 enters the fetal circulation. Finally, similar vascular alterations were observed in human placentas from pregnancies with severe maternal inflammation. These findings reveal a conserved inflammatory mechanism that compromises PBB integrity and may relay signals to the fetal brain, highlighting potential therapeutic targets for neuroprotection during prenatal inflammation.