A Unifying Thermodynamic Model for Phase Separation and Aging of Biopolymers
A Unifying Thermodynamic Model for Phase Separation and Aging of Biopolymers
Michels, J. J.; Caria, J.; Lemke, E. A.
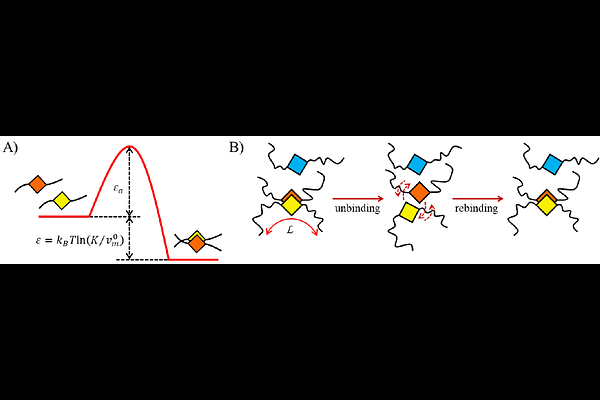
AbstractProtein condensates that form via phase separation typically become more viscous over time and can harden in a process referred to as 'molecular aging'. Several mechanisms have been identified for this phenomenon. Of these, the ones involving enhanced {beta}-sheet or -strand interactions are of pathological relevance since they have been associated with neurodegeneration. Although there is much understanding of biopolymer phase behavior, an inclusive thermodynamic framework that unifies phase separation and {beta}-sheet-based aging is lacking. We present a time-dependent, multi-component extension of associating polymer theory that describes phase separation and aging of an intrinsically disordered protein (IDP) capable of associating through local, reversible folding. The model shows how the Second Law of Thermodynamics applies throughout, whether phase separation precedes and encourages aging or, vice versa, whether the increase in 'stickiness' during aging drives phase separation. Our calculations show how the time-dependence of the average valency of associating sites determines the aging kinetics and the development of viscoelastic properties of a biocondensate. The agreement between our calculations and the change in dynamics of condensates of 'perfect repeat' analogues of nucleoporin-98 not only validates the theory but also identifies these Nup98 variants as model systems for studying aging.