Lateral hypothalamic input engages a disinhibitory microcircuit in the dorsal raphe to promote behavior activation
Lateral hypothalamic input engages a disinhibitory microcircuit in the dorsal raphe to promote behavior activation
Sadretdinova, R.; Rivera, A. M.; Benmamma, Z.; Pinel, M.; Emond, V.; Zhao, C.; Calon, F.; Labonte, B.; Proulx, C. D.
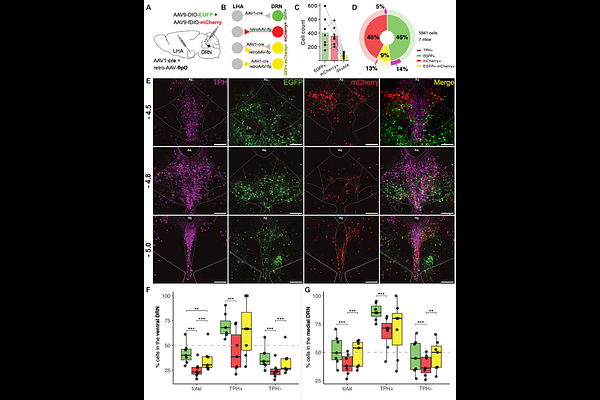
AbstractBehavioral activation involves coordination between hypothalamic and brainstem systems that regulate movement and internal state, but the circuit logic underlying this interaction remains poorly defined. The dorsal raphe nucleus (DRN), a major serotonergic hub, integrates diverse inputs to influence behavioral inhibition and activation, yet how hypothalamic signals shape its activity is unclear. Here, we identify a disinhibitory pathway through which the lateral hypothalamus (LHA) promotes behavioral activation via DRN. Using intersectional viral tracing, electrophysiology, and single-nucleus RNA sequencing, we show that LHA inputs preferentially target GABAergic DRN neurons that locally inhibit 5-HT neurons. Silencing these DRN neurons innervated by LHA increased locomotor and repetitive behaviors, decreased local inhibition, and enhanced cFos activity in serotonergic neurons, consistent with circuit-level disinhibition. Molecular profiling revealed distinct transmitter identities and transcriptional signatures of LHA-targeted versus LHA-projecting DRN populations. Together, these findings delineate a hypothalamic-raphe circuit that transforms hypothalamic drive into serotonergic activation, revealing a mechanism by which the lateral hypothalamus promotes behavioral activation through local inhibitory control.