Rare variants alter mitochondrial lipid homeostasis and neuronal excitability in PD patient-derived dopaminergic neurons
Rare variants alter mitochondrial lipid homeostasis and neuronal excitability in PD patient-derived dopaminergic neurons
Esposito, T.; Carrillo, F.; Fortunato, G.; Coppola, A.; Ghirimoldi, M.; Okechukwu, N. G.; Borrini, V. F.; Khoso, S.; Di Lorenzo, A.; Marciano, M.; Giurin, G.; D'Amato, F.; Iazzetta, M. R.; D'Aniello, C.; Fiorenzano, A.; Nutile, T.; Licastro, D.; Pietracupa, S.; Modugno, N.; Martinello, K.; Fucile, S.; Manfredi, M.; Fico, A.
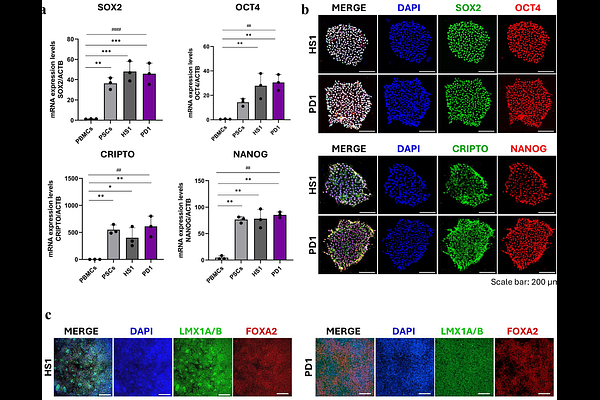
AbstractParkinson's disease (PD) exhibits substantial genetic heterogeneity, yet how combinations of rare variants converge on disease-relevant cellular mechanisms remains unclear. Here, we generated human induced pluripotent stem cell-derived dopaminergic neurons from PD patients carrying rare variants in recently implicated genes and performed integrated electrophysiological, proteomic, lipidomic, and genetic analyses. Patient-derived neurons showed reduced membrane capacitance and altered action potential firing, indicating impaired intrinsic excitability and synaptic dysfunction, with marked variability across genetic backgrounds. Multi-omics profiling revealed dysregulation of mitochondrial function, glycolysis, and oxidative phosphorylation, accompanied by extensive lipid remodeling, including increased fatty acids, acylcarnitines, and sphingolipids, and reduced gangliosides. These alterations were more pronounced in neurons harboring specific variant combinations in KIF21B, SLC6A3, HMOX2, TMEM175, and AIMP2. Integrative analyses uncovered coordinated protein-lipid changes linking mitochondrial dysfunction and membrane homeostasis. Notably, Calpastatin and CXCR4 were consistently dysregulated across PD neurons. Genetic association analyses in independent cohorts identified PD-associated variants in genes encoding dysregulated proteins, supporting the functional relevance of these pathways. Overall, our results define convergent and variant-specific mechanisms underlying PD and highlight candidate biomarkers and therapeutic targets.