Integrated CHARGE syndrome models reveal epigenetic modulators of reproductive phenotypes
Integrated CHARGE syndrome models reveal epigenetic modulators of reproductive phenotypes
Amoruso, F.; La Rocca, F.; Santonicola, P.; Paganoni, A. J. J.; Zampi, G.; Manzini, S.; Fontana, F.; Cristofani, R.; Oleari, R.; Di Schiavi, E.; Cariboni, A.
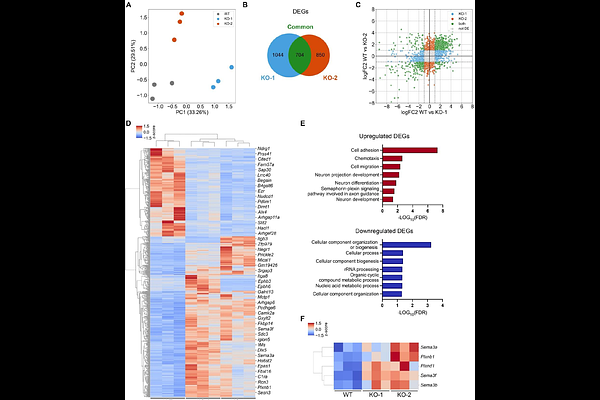
AbstractLoss-of-function variants in CHD7 cause CHARGE syndrome (CS), a rare developmental disorder showing multisystem malformations, including reproductive defects linked to gonadotropin-releasing hormone (GnRH) neuron dysfunction. CHD7 encodes a chromatin remodeler essential for early transcriptional regulation across various tissues. Currently, no pharmacological treatments exist, and approaches aimed at identifying tissue-specific CHD7 targets are also lacking, making CS treatment an unmet clinical need. To explore mechanisms relevant to CS-associated reproductive defects, we established a dual screening platform combining CRISPR-engineered Chd7-depleted mouse GnRH neurons with a Caenorhabditis elegans chd-7 mutant showing reproductive abnormalities. Transcriptomic and functional analyses of Chd7-deficient cells revealed impaired cellular processes along with dysregulation of semaphorin (Sema) genes, key regulators of GnRH neuron development. A screen of 234 epigenetic modulators in C. elegans identified compounds modifying aberrant mutant phenotype, two of which also rescued cellular defects and Sema expression in vitro. Altogether, these findings indicate that CHD7 deficiency reshapes neuroendocrine-relevant pathways and that selected compounds modulate CS-associated phenotypes across species, with SEMA signalling emerging as candidate druggable downstream pathway in CS requiring further mechanistic validation.