Lung-Selective Immune Reprogramming via In Situ Red Blood Cell Hitchhiking Nanoparticles
Lung-Selective Immune Reprogramming via In Situ Red Blood Cell Hitchhiking Nanoparticles
Udofa, E. M.; Zhang, E.; Naderi, M. M.; He, S.; Gunasekara, H.; Feng, B.; Chao, C.-J.; Lee, J.; Herrera-Alonso, M.; Liu, Y.; Hu, Y. S.; Peng, Z.; Zhao, Z.
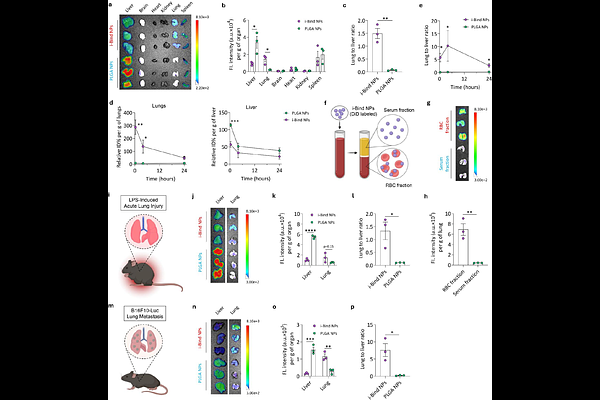
AbstractPremature clearance and limited organ targeting remain major barriers for nanoparticle (NP) drug delivery. Hitchhiking NPs on red blood cells (RBCs) can enhance circulation and organ-selective accumulation, but most approaches require ex vivo RBC extraction and reinfusion, limiting clinical translation. Here, we report an in situ RBC-hitchhiking strategy, named i-Bind, which employs polyphenol surface functionalization to enable spontaneous NP attachment to RBCs directly in the bloodstream. Driven by strong interactions of phenolic motifs with RBC membranes, i-Bind NPs exhibited markedly enhanced and more stable hitchhiking onto RBCs under flowing whole blood conditions. In both healthy and diseased mice, i-Bind NPs selectively target the lungs, resulting in an over 20-fold increase in lung-to-liver deposition ratio compared to unmodified NPs. Additionally, i-Bind NPs show preferential targeting to distinct lung immune cell subsets in a pathology-dependent manner, including cDC2s in healthy lungs, neutrophils in acute lung injury, and cDC1s in lung metastases. In a melanoma lung metastasis model, delivery of the STING agonist diABZI via i-Bind NPs significantly inhibited lung metastasis progression by reprogramming the lung immune microenvironment. Collectively, i-Bind provides a simple and versatile platform for organ-selective drug delivery and immune reprogramming.