Title: Multimodal profiling reveals Mycobacterium tuberculosis restricts lung mitochondrial immunometabolism to promote pathogenesis
Title: Multimodal profiling reveals Mycobacterium tuberculosis restricts lung mitochondrial immunometabolism to promote pathogenesis
Dkhar, H. K.; Bajpai, P.; Enriquez, A. B.; Hopkins, L. B.; Dawa, S.; Sia, J. K.; Paul, A.; Madan-Lala, R.; Keenum, M. C.; Sharma, A.; Pelletier, A. N.; Goh, S. L.; Riddick, T. L. B.; Whitworth, T. J.; Prater, K. E.; Guerrero-Ferreira, R. C.; Collins, J. M.; Rengarajan, J.
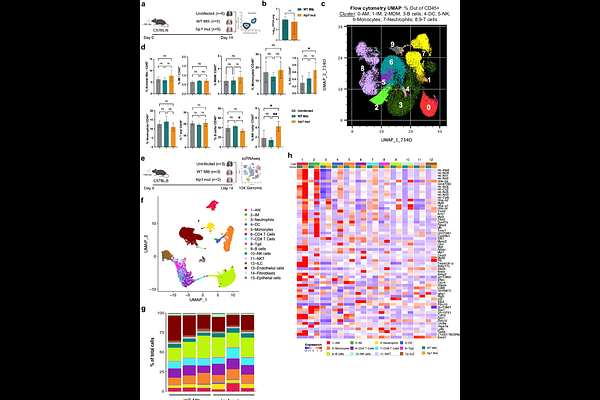
AbstractEarly events in the lung that shape protective immune responses to M. tuberculosis (Mtb) infection are not well understood but are critical for developing better vaccines and immunomodulatory therapies for tuberculosis. Here, we used high-dimensional flow cytometry, single-cell transcriptomics, and untargeted metabolomics to define the early lung immune environment that precedes the development of protective versus pathogenic outcomes following aerosolized Mtb infection of mice. We show that Mtb induced sustained glycolysis in the lung while restricting oxidative phosphorylation (OXPHOS) and impairing mitochondria, in part through the Mtb serine protease Hip1, leading to low energy output and suboptimal macrophage-T cell interactions that promoted pathogenic immunity. However, robust induction of mitochondrial OXPHOS, amino acid metabolism, and fatty acid oxidation in the early lung resulted in high ATP output and enhanced innate-T cell signaling networks that stimulated protective immune responses. Moreover, we identified a novel mitochondrial immunometabolic lung signature associated with protective outcomes to Mtb infection in animal models and humans. Our studies identify induction of mitochondrial dysfunction as a mechanism employed by Mtb to manipulate lung immunometabolism to its benefit and reveal that maintenance of intact mitochondrial metabolism in the early lung is pivotal for generating protective outcomes to Mtb infection.