immunoPETE: A DNA-based integrated B-cell and T-cell receptor profiling platform
immunoPETE: A DNA-based integrated B-cell and T-cell receptor profiling platform
Zhao, H.; Mirebrahim, H.; Telman, D.; Dannebaum, R.; McNamara, S.; Tabari, E.; Lin, H.; Rubelt, F.; Berka, J.; Luong, K.; Joseph, M.; Bryan, R.; Ward, D.; Hayday, A.; Utiramerur, S.; Kumar, D.; Asgharian, H.
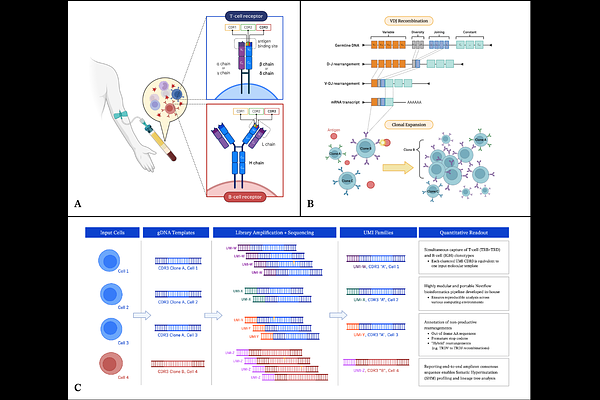
AbstractThe vast diversity of B and T cell receptors generated through the recombination of Variable (V), Diversity (D), and Joining (J) gene segments plays a critical role in adaptive immunity. Profiling immune repertoires at the DNA level provides a robust and stable approach to capture the clonal composition of these receptors. immunoPETE is an assay designed to target recombined human T-cell Receptor Beta (TRB), T-cell Receptor Delta (TRD), and Immunoglobulin Heavy (IGH) chain genes directly from genomic DNA. Simultaneous profiling of B and T cell receptor chains in a single reaction provides internally normalized clone counts and facilitates the study of B-T cell interactions. Full-length amplicon consensus sequences representative of original template DNA molecules are accurately reconstructed using Unique Molecular Identifiers (UMIs). An in-house pipeline compiles VDJ rearrangements from the Complementarity-Determining Region 3 (CDR3) of TRB, TRD and IGH chains into comprehensive readouts at cell-level resolution. In this study, we describe the immunoPETE end-to-end workflow, followed by a comprehensive benchmarking of its performance in adaptive immune profiling. Where applicable, we used both natural and contrived samples and characterized the assay's accuracy, linearity, and reproducibility across several metrics: retrieving CDR3 sequences, determining B and T cell ratios, total cell count, yield, fraction of functional rearrangements, clonal diversity, composition of dominant clones, pairwise similarity, and V/J gene usage frequencies. Furthermore, we assessed its quantitative limits concerning the total number of lymphocytes and the detection of rare clones. As an example of its applications, we show that adding immune biomarkers extracted from immunoPETE data to clinical factors improves prediction of progression-free survival in a cohort of non-muscle invasive bladder cancer (NMIBC) patients. Finally, we discuss the broad applications of immunoPETE in the study of aging, cancers, infections, and autoimmune disorders with reference to select published studies.