Aging-associated endo-lysosomal dysfunction drives inflammaging and neurodegeneration through the STING-IFN-I axis
Aging-associated endo-lysosomal dysfunction drives inflammaging and neurodegeneration through the STING-IFN-I axis
Oberg, M.; Maric, I. P.; Stromberg, A.; Myers, C.; Saffarzadeh, N.; Fabrikova, D.; Fabrik, I.; Rivas Gavalez, L.; Skibicka, K.; Kurzawa-Akanbi, M.; Paul, G.; Gekara, N. O.; Hartlova, A.
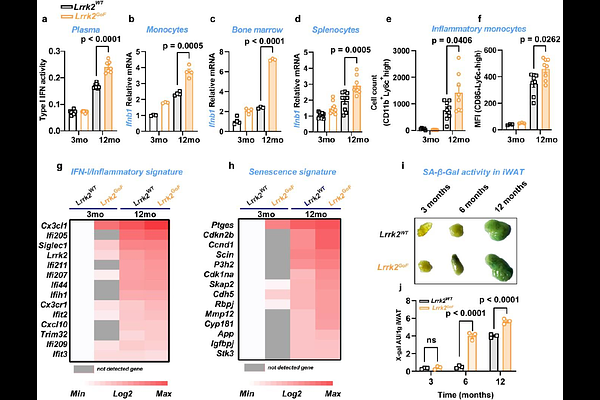
AbstractAll animals age. However, aging is a heterogeneous process, and individual organisms age differently. Moreover, within the same organism, cells or organs do not age at the same time or speed. For instance, although neurodegeneration is a key trait of aging, neurological symptoms normally manifest long after multiple indicators of aging in peripheral tissues. The genetic determinants of aging remain poorly understood. Mutations in leucine-rich repeat kinase 2 (LRRK2) are major genetic risk factors for Parkinson disease (PD). By analyzing PD patients and mice with an LRRK2 gain-of-function mutation (LRRK2GoF), we demonstrate that PD is an accelerated aging disease characterized by systemic low-grade STING-dependent inflammation (inflammaging) that first manifests in the periphery, then disrupts the blood-brain barrier and progresses to the brain, resulting in neurodegeneration. Mechanistically, we demonstrate that a primary consequence of aging or Lrrk2GoF is endolysosomal decline. This results in the cytosolic build-up of extraneous self-DNA and subsequent shedding of DNA-containing extracellular vesicles, thereby triggering the cGAS-STING pathway cell-intrinsically and intercellularly in distant host cells. This study unveils the cGAS-STING pathway and LRRK2GoF as key determinants and potential targets for preventive or therapeutic strategies against accelerated aging, inflammaging, and neurodegeneration.