Developmental Synchrony of Retinal Waves, Apoptosis, and Angiogenesis in Postnatal Retina
Developmental Synchrony of Retinal Waves, Apoptosis, and Angiogenesis in Postnatal Retina
Savage, M. A.; Bertram, C.; de Montigny, J.; Thorne, C. A.; Queen, R.; Lako, M.; Hilgen, G.; Sernagor, E.
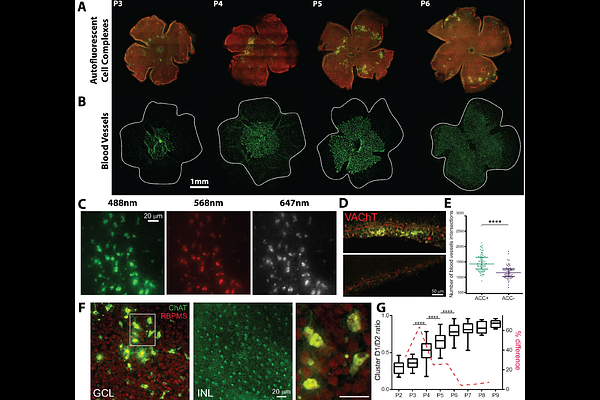
AbstractPostnatal mouse retinal development is a multi-faceted process involving the coordinated interaction of spontaneous neural activity as retinal waves, vascular plexus growth, and programmed cell death. While these processes are known to interact at a coarse scale, the specific mechanisms integrating them have remained elusive. Using large-scale, widefield calcium imaging, high-density multielectrode array recordings, single cell RNA-seq, and immunohistochemistry, we characterise a tightly aligned centrifugal expansion pattern during retinal development. This pattern is common to stage II retinal wave onsets, vascular development, Heme oxygenase-1 (Hmox1) expressing microglia, apoptotic cell markers, and a novel set of auto-fluorescent cluster complexes (ACCs) identified in this study. Apoptotic cells are known to upregulate functional Pannexin1 (PANX1) hemichannels. These voltage-gated channels release purinergic molecules which act as "eat me" signals to neighbouring microglia. PANX1 hemichannel blockade with the drug probenecid results in a profound decrease in spontaneous wave frequency and strength, suggesting that retinal waves are indeed triggered by these apoptotic cells. Taken together, our observations suggest that spontaneous waves are initially triggered in hotspots by hyperactive apoptotic RGCs in unvascularised retinal areas. These apoptotic cells release purinergic molecules via PANX1 hemichannels, leading to wave generation. This hyperactivity leads to local hypoxic conditions, which, coupled with high extracellular ATP concentrations, promotes angiogenesis. Once blood vessels reach a particular hotspot, ATP release activates Hmox1 positive microglia, which engulf the dying RGCs, creating the auto-fluorescent clusters. We suggest that these early occurring events may be universal in the developing CNS, causally linking early neural activity, programmed cell death and angiogenesis.