A centromere but not just a centromere: structure and evolution of a selfish chromosomal supergene in monkeyflowers
A centromere but not just a centromere: structure and evolution of a selfish chromosomal supergene in monkeyflowers
Stark-Dykema, E.; Finseth, F. R.; Conner, W. R.; Fishman, L.
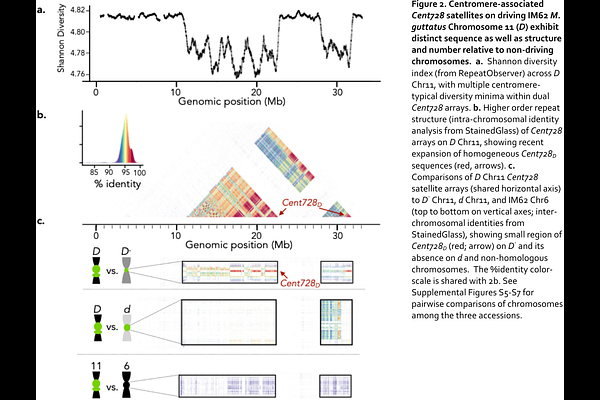
AbstractMeiotic drivers capitalize on vulnerabilities in eukaryotic meiosis and gametogenesis to gain greater-than-equal transmission to the next generation. In most plants and animals, asymmetric female meiosis is such an arena for selfishness; centromeric variants can bias their chromosomes segregation to the one egg cell with potentially profound effects on individual fitness, population dynamics, and genome structure. However, the mechanisms and consequences of selfish centromere evolution remain obscure, due to both the complexity of centromeric DNA and to a paucity of model systems. Here, we build on representative chromosome-scale assemblies of yellow monkeyflowers (M. guttatus species complex) to compare the structure, gene content, and sequence of three functionally distinct haplotypes at Meiotic Drive Locus on Chromosome 11 (MDL11), whose driving D allele was previously shown to be centromere-containing, costly, and maintained as a balanced polymorphism in wild populations. Relative to both moderately-resistant M. guttatus nondrivers (D-; IM767) and weak (d)M. nasutus, D minimally evolved via two rearrangements (including a hemicentric inversion creating dual arrays of M. guttatus centromeric satellite Cent728 separated by > 200 trapped genes), massive expansions of a novel MDL11-specific subfamily of satellite repeats (Cent728D) that increase D chromosome length by >50%, and the accumulation of >40 extra D-only genes (EDG) from diverse genomic sources in a hemizygous pericentromeric region. Under a strictly centromeric model of selfishness, the recent expansion of Cent728D is a primary candidate for generating centromere drive, either by directly altering the binding of centromeric histone CenH3 or by shifting kinetochore size or position as an epigenetic byproduct of chromosome size. However, elevated divergence in genic presence-absence variants (both in the EDG and elsewhere) and gene sequence across MDL11 strata points to strong candidates for both collusion with the D centromere and for interspecific (D- vs. d) variation in resistance. Overall, our findings support the sequential buildup of this model centromeric driver over time, reveal intriguing parallels with adaptive and gamete-killing supergenes, and provide a strong platform for further dissection of the molecular genetic basis of its dramatic deviations from Mendelian rules.